 |
市場調查報告書
商品編碼
1958586
互聯藥物傳輸設備市場(第三版):依設備類型、連接方式、給藥途徑、治療領域和地區劃分 - 趨勢與至2035年的預測Connected Drug Delivery Devices Market (3rd Edition): Distribution by Type of Device, Mode of Connectivity, Route of Administration, Therapeutic Area & Geographical Regions - Trends and Forecast Till 2035 |
||||||
互聯藥物傳輸設備市場 - 概述
全球互聯藥物輸送設備市場預計將從目前的53億美元成長到2035年的127億美元,在預測期內(至2035年)的年複合成長率(CAGR)為 10.2%。
互聯藥物傳輸設備市場 - 成長與趨勢
藥物依從性低一直是醫療保健領域的重大挑戰,它會導致疾病進展、住院率上升、死亡率升高以及醫療保健成本增加。研究表明,儘管美國約有 66%的人至少獲得過一次處方,但約有 50%的人未能按醫囑服藥。藥物依從性問題源自於多種因素,包括複雜的給藥方案、藥物取得途徑有限、病患支持不足。為了應對這項挑戰,醫療保健專業人員和技術提供者採用各種策略,包括簡化給藥方案、提供個人化給藥計畫以及加強病患教育。
互聯藥物輸送設備透過將傳統給藥方式與數位健康技術結合,成為提高用藥依從性的有效解決方案。這些設備配備了先進的功能、感測器和無線連接,能夠與行動應用程式、數位健康平台和電子健康記錄(EHR)無縫整合。此外,互聯藥物輸送系統能夠即時追蹤藥物攝取量和患者健康狀況,促進及時的臨床干預和個人化治療方案的調整。透過提高患者參與度和依從性,這些設備不僅能夠改善治療效果,還有助於降低整體醫療保健成本。
此外,感測器技術的整合改變傳統藥物傳輸系統的功能,使其發展成為智慧互聯的解決方案。遠距醫療整合的持續進步和數位健康工具的廣泛應用預計將在未來幾年推動互聯藥物輸送設備市場持續成長。
成長因素 - 市場擴張的策略驅動因素
糖尿病、氣喘、慢性阻塞性肺病(COPD)和自體免疫疾病等慢性病盛行率的上升推動市場需求。這是因為互聯藥物傳輸設備能夠實現精準的劑量管理和長期治療管理。感測器技術、低功耗藍牙、人工智慧分析和物聯網整合等方面的技術進步,使得遠端監測、與醫療服務提供者的資料共享以及個人化干預成為可能,提高了患者的用藥依從性和治療效果。患者參與度的提高和人口老化進一步推動了市場成長,而與遠距醫療的協同作用以及向精準醫療的轉變,預計將推動市場實現更高的年複合成長率。
市場挑戰 - 發展道路上的重大障礙
互聯藥物輸送設備整合了感測器和應用程序,可實現對糖尿病和哮喘等慢性疾病的即時用藥依從性追蹤和遠端監測,但其面臨著多方面的市場挑戰,阻礙了其廣泛應用。由於先進的感測器、研發、精密組裝和供應鏈問題,高昂的開發和製造成本推高了其價格,使其高於傳統設備。此外,持續的雲端服務費用、關稅和償付缺口也加劇了這些挑戰。此外,藥物-設備組合的嚴格監管障礙和緩慢的全球合規進程與快速的技術進步相衝突,限制了人工智慧的創新。網路安全和資料隱私風險削弱了用戶信任,並限制了資料共享。此外,可用性問題等應用障礙依然存在。
互聯藥物傳輸設備市場 - 關鍵洞察
本報告深入分析了互聯藥物輸送設備市場的現狀,並指出了該行業的潛在成長機會。主要發現包括:
- 在當前市場格局中,超過 80 家製造商提供各種互聯藥物輸送設備,其中大多數總部位於北美。
- 目前約有 75%的互聯/智慧藥物輸送設備已上市,其中大部分設備用於皮下給藥。

- 目前,超過 30 種可與傳統藥物輸送設備整合的創新附加感測器已上市或研發中。這些附加感測器製造商大多是位於歐洲的新創公司。
- 超過 55%的附加感測器專為吸入式藥物輸送而設計,其中超過 85%的感測器支援藍牙連接。
- 迄今為止,該領域註冊的大多數臨床試驗都在評估各種連網藥物傳輸裝置的安全性和有效性,其中許多研究在美國各地多個地點進行。
- 近年來,各利害關係人之間的合作關係日益增多,反映出利害關係人對該市場的興趣日益濃厚。事實上,近 70%的交易都是在過去三年內達成的。
- 投資者正向連網藥物輸送裝置市場投入大量資金,以開發能加速產業發展的先進設備。
- 連網/智慧型藥物傳輸裝置的開發過程包括多個階段,包括規劃、設計、工程、製程驗證、製造、組裝和商業化。
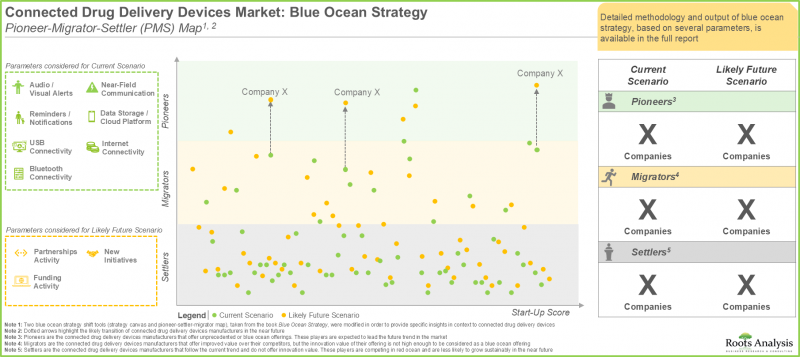
- 根據藍海戰略分析中的 "先鋒-遷徙-定居者" 地圖,未來幾年,多家專注於產品改進的互聯藥物輸送設備製造商有望成為行業先鋒。
- 由於互聯藥物輸送設備能夠實現精準給藥和遠端患者監測,預計未來十年該市場將實現顯著成長(年複合成長率達10.2%)。
- 預計到2035年,北美互聯藥物輸送設備市場將佔據最大佔有率。此外,大多數用於藥物傳輸的設備都針對代謝性疾病。
- 受美國智慧互聯藥物輸送解決方案日益普及的推動,預計互聯藥物輸送設備市場在預測期內將以更高的年複合成長率成長。
- 鑑於對先進藥物輸送設備的需求激增以及慢性病患病率的上升,互聯藥物輸送設備市場預計將穩步擴張。
互聯藥物傳輸設備市場
市場規模和機會分析基於以下參數進行細分:
依裝置類型
- 連網/智慧型穿戴注射器
- 互聯/智慧吸入器
- 互聯/智慧注射筆
- 連網/智慧型自動注射器
- 互聯/智慧型無針注射系統
依連接方式
- 藍牙設備
- 近場通訊設備
- 蜂巢式網路設備
- 廣域網路設備
依給藥途徑
- 腸外給藥
- 吸入給藥
依治療領域
- 代謝性疾病
- 呼吸系統疾病
- 自體免疫疾病
- 其他疾病
依地區
- 北美洲
- 美國
- 加拿大
- 歐洲
- 英國
- 德國
- 法國
- 義大利
- 西班牙
- 歐洲其他地區
- 亞太地區
- 中國
- 印度
- 日本
- 亞太其他地區
互聯藥物輸送設備市場 - 主要細分市場
連網/智慧型穿戴注射器佔據最大市場佔有率
連網/智慧型穿戴注射器今年佔據了最大的市場佔有率,預計這一趨勢將在未來幾年持續下去。這主要歸因於慢性病盛行率的不斷上升,以及利害關係人持續致力於將先進功能融入連網藥物輸送設備。這些改進能夠增強監測效果、提高病患依從性,並促進與醫療專業人員的資料交換。
未來藍牙連網/智慧型裝置可望佔最大市場佔有率
目前,藍牙連網/智慧型裝置預計將佔據連網藥物傳輸設備市場更大的佔有率。此外,預計該細分市場在預測期內將實現顯著成長。這是因為藍牙連接易於設置,幾乎所有智慧型手機都支援此功能,而且無需額外設備即可運作。
今年,腸外給藥領域將佔據連網藥物輸送設備市場最大的佔有率。
據估計,腸外給藥領域今年的市場佔有率預計將更大。這是因為腸外給藥是生物製劑和慢性病高價值療法的首選給藥途徑。此外,吸入給藥領域預計在預測期內將維持較高的年複合成長率。這主要歸功於吸入給藥方式的便利性、多功能性和無痛性,以及其能夠提供持續的局部治療效果。
北美將佔據市場主導地位,擁有最大的市場佔有率。
根據預測,北美預計在今年繼續保持其在互聯藥物輸送設備市場佔有率方面的領先地位。此外,預計到2035年,亞太市場將以相對較高的成長率擴張。這一成長得益於生物製藥新創企業的增加、創新藥物輸送系統研發的加強以及該地區人口的成長。這主要得益於互聯藥物輸送設備市場中獲準用於治療各種疾病的設備數量的增加。
互聯藥物輸送設備市場的主要參與者
- Bigfoot Biomedical
- BIOCORP
- Eitan Medical
- E3D Elcam Drug Delivery Device
- EoFlow
- Gerresheimer
- Insulet
- Medtronic
- Medtrum
- Nemera
- Phillips-Medisize
- Roche
- Sonceboz
- SOOIL Development
- Tandem Diabetes Care
- West Pharmaceutical Services
- Ypsomed
互聯藥物傳輸設備市場 - 範圍
- 市場規模和機會分析:本報告對互聯藥物輸送設備市場進行了詳細分析。 藥物輸送裝置市場,重點關注以下關鍵細分市場:[A] 裝置類型,[B] 連接方式,[C] 給藥途徑,[D] 治療領域,以及 [E] 地理區域。
- 互聯/智慧型藥物輸送裝置市場概覽:除了對整合式互聯藥物輸送裝置的整體市場概覽進行詳細概述外,還提供以下資訊:[A] 開發階段,[B] 裝置上市年份,[C] 整合裝置類型,[D] 給藥途徑,[E] 目標適應症/治療領域,[F] 整合裝置類型,[D] 給藥途徑,[E] 目標適應症/治療領域,[F] 主要功能,[G] 平台的應用方式,[成立年份,[J] 公司規模,[K] 總部所在地,以及 [H] 主要參與者。
- 附加感測器市場概覽:本報告概述了附加感測器市場的整體市場格局,並詳細介紹了以下相關參數:[A] 開發階段,[B] 設備發布年份,[C] 相容設備,[D] 給藥途徑,[E] 目標適應症/治療領域,[F] 治療領域,[G] 目標可適應症,[E] 目標適應症/治療領域,[F] 治療領域,[G] 目標可適應症,[HJ] 主要可用性,[KJ]資料儲存/雲端平台的可用性,[L] 成立年份,[M] 公司規模,[N] 總部所在地,[O] 主要參與者。
- 產品競爭分析:本報告基於 [A] 製造商優勢和 [B] 產品組合優勢,對互聯藥物輸送設備和附加感測器進行調查分析。
- 公司概況:本報告基於以下幾個方面,詳細介紹了北美、歐洲和亞太地區主要互聯藥物輸送設備製造商的概況:[A] 成立年份,[B] 總部所在地,[C] 產品組合,[D] 近期發展,以及 [E] 未來展望。
- 臨床試驗分析:基於以下幾個相關參數,分析了已完成和進行的與互聯藥物輸送設備相關的臨床試驗:[A] 研究註冊年份,[B] 入組患者人數,[C] 研究狀態,[D] 申辦方/合作方類型,[E] 研究設計,以及 [F] 所在地區。
- 合作關係:基於以下相關參數,分析了利害關係人之間達成的合作關係:[A] 合作年份,[B] 合作類型,[C] 設備分類,[D] 整合設備類型,以及 [E] 最活躍的參與者。
- 資金/投資分析:基於以下相關參數分析該領域的資金和投資協議:[A] 資金年份,[B] 資金類型,[C] 投資金額,[D] 地區,以及 [E] 主要參與者。
- 價值鏈分析:提供連網/智慧藥物輸送裝置開發各階段的詳細價值鏈分析,例如研發、產品製造/組裝、產品分銷、行銷/銷售以及上市後監測,並提供各階段的成本資訊。
- 監管環境:概述各國家和地區主要監管機構制定和發布的互聯藥物輸送裝置批准指南。
- 案例研究:針對兩種最常見適應症(即氣喘和糖尿病)的詳細案例研究,包括疾病概述、流行病學、經濟負擔(因未遵醫囑治療而造成的經濟負擔),以及藥物輸送裝置的發展歷程和其他目的是提高這些適應症患者治療依從性的策略概述。
- 藍海策略分析:基於藍海策略,對當前及未來市場進行詳細分析。本報告為新興的互聯藥物輸送設備公司提供策略規劃/指南,幫助他們開拓未開發的市場,並提供策略工具,協助其轉型藍海策略,在市場中獲得競爭優勢。
- SWOT 分析:透過 SWOT 分析,深入探討可能影響連網藥物輸送設備市場發展的產業相關趨勢、機會與挑戰。
目錄
第1章 引言
第2章 研究方法
第3章 市場動態
第4章 宏觀經濟指標
第5章 執行摘要
第6章 引言
- 章節概述
- 藥物依從性
- 智慧型/互聯藥物輸送裝置概述
- 關鍵設計特性與開發流程
- 智慧型/互聯藥物傳輸裝置的分類
- 智慧型/互聯藥物輸送裝置的監管指南
- 智慧型/互聯藥物傳輸裝置的優點和局限性
- 新冠疫情對智慧/連網藥物輸送裝置的影響產業
- 未來展望
第7章 市場概況:互聯/智慧型藥物傳輸設備
- 章節概述
- 互聯/智慧型藥物傳輸設備:市場概況
- 互聯/智慧型藥物傳輸設備製造商:市場概況
第8章 市場概況:附加感知器
- 章節概述
- 附加感測器:市場概況
- 附加感測器製造商:市場概況
第9章 產品競爭分析
- 章節概述
- 假設和關鍵參數
- 研究方法
- 互聯藥物輸送設備與附加感測器:產品競爭分析
第10章 公司簡介:北方美國互聯/智慧藥物傳輸設備製造商
- 章節概述
- 主要互聯/智慧型藥物傳輸設備製造商
- Medtronic
- Insulet
- Tandem Diabetes Care
- 其他廠商
- Bigfoot Biomedical
- Phillips Medisize(Molex旗下子公司)
- West Pharmaceutical Services
第11章 公司簡介:歐洲互聯/智慧藥物傳輸設備製造商
- 章節概述
- 主要互聯/智慧型藥物傳輸設備製造商
- BIOCORP
- Gerresheimer
- Roche
- Ypsomed
- 其他廠商
- Nemera
- Sonceboz
第12章 公司簡介:亞太及其他地區互聯/智慧藥物傳輸設備製造商
- 章節概述
- 主要互聯/智慧型藥物傳輸設備製造商
- Medtrum
- SOOIL Development
- EOFlow
- 其他廠商
- Eitan Medical
- E3D Elcam Drug Delivery設備
第13章 臨床試驗分析
- 章節概述
- 研究範圍與方法
- 互聯/智慧型藥物傳輸設備和附加感測器:臨床試驗分析
第14章 夥伴關係與合作
- 章節概述
- 合作模式
- 互聯/智慧型藥物傳輸設備:夥伴關係與合作
第15章 資金與投資分析
- 章節概述
- 資金模式
- 互聯/智慧藥物輸送設備:資金與投資
第16章 價值鏈分析
- 章節概述
- 互聯/智慧型藥物輸送裝置:價值鏈
- 價值鏈成本分配
第17章 互聯/智慧型藥物輸送裝置的監管環境
- 章節概述
- 北美組合產品的監理審批
- 美國組合產品的監管審批
- 加拿大組合產品的監理審批
- 墨西哥組合產品的監管審批
- 歐洲組合產品的監理審批
- 歐盟五國組合產品的監管審批
- 亞太地區組合產品的監管審批
- 日本組合產品的監管核准
- 中國組合產品的監管核准
- 印度組合產品的監管核准
- 韓國組合產品的監管核准
- 澳洲組合產品的監管批准
- 中東和北非組合產品的監管批准
- 沙烏地阿拉伯組合產品的監管核准
- 阿拉伯聯合大公國組合產品的監管批准
- 拉丁美洲組合產品的監管批准
- 巴西組合產品的監管核准
- 阿根廷組合產品的監管批准
- 連網設備:其他安全、有效性和效能
- 確保收集資料的安全
- 結論
第18章 案例研究:應用互聯藥物輸送裝置提高用藥依從性
- 章節概述
- 個案研究1:糖尿病
- 案例研究2:氣喘
第19章 SWOT分析
第20章 藍海策略
- 藍海策略概述
第21章 全球互聯藥物輸送設備市場
第22章 互聯藥物輸送設備市場(依設備類型劃分)
第23章 互聯藥物輸送設備市場(以連接方式劃分)
第24章 互聯藥物輸送設備市場(依給藥途徑劃分)
第25章 互聯藥物輸送設備市場(依治療領域劃分)
第26章 互聯藥物輸送設備市場(依地區劃分)
第27章 互聯藥物輸送設備市場(依主要廠商劃分)
第28 結論
第29章 高層洞察
第30章 附錄1:表格資料
第31章 附錄2:公司與組織清單
第32章 附錄3:2015-2020年合作夥伴關係
Connected Drug Delivery Devices Market: Overview
As per Roots Analysis, the global connected drug delivery market is estimated to grow from USD 5.3 billion in the current year to USD 12.7 billion by 2035, at a CAGR of 10.2% during the forecast period, till 2035.
Connected Drug Delivery Devices Market: Growth and Trends
Over the years, poor compliance to prescribed medications continues to be a significant issue in the healthcare field, leading to disease progression, increased hospitalization rates, mortality, and rising healthcare expenses. Studies indicate that approximately 66% of individuals in the United States receive at least one prescription, yet nearly 50% fail to adhere to their physicians' instructions for medication use. This challenge of non-adherence arises from multiple factors, such as complicated dosing regimens, restricted access to medications, and inadequate patient assistance. To address this, healthcare professionals and technology providers are using various strategies to streamline dosage plans, provide individualized medication schedules, and improve patient education.
Connected drug delivery devices represent a viable solution as these devices integrate traditional delivery methods with digital health technologies to improve adherence. These devices come with advanced features, sensors, and wireless connectivity that allow for smooth integration with mobile apps, digital health platforms, and electronic health records. Additionally, the linked drug delivery system allows for real-time tracking of medication consumption and patient health conditions, thus facilitating prompt clinical interventions and customized therapy modifications. By enhancing patient involvement and adherence, these devices not only bolster patient results but also help in reducing total healthcare costs.
Further, the integration of sensor technologies has transformed the capabilities of conventional drug delivery systems, allowing them to evolve into intelligent, interconnected solutions. Ongoing advancements in telehealth integration and broader acceptance of digital health tools are anticipated to fuel consistent growth in the connected drug delivery device market in the upcoming years.
Growth Drivers: Strategic Enablers of Market Expansion
Rising chronic disease prevalence, including diabetes, asthma, chronic obstructive pulmonary disease (COPD), and autoimmune disorders, fuels demand. This is due to the fact that connected drug delivery devices enable precise dosing and long-term therapy management. Technological advances in sensors, Bluetooth Low Energy, AI analytics, and IoT integration allow remote monitoring, data sharing with providers, and personalized interventions, boosting adherence and outcomes. Patient engagement and aging populations further accelerate growth, with market projections showing a higher CAGR driven by telehealth synergies and precision medicine shifts.
Market Challenges: Critical Barriers Impeding Progress
Connected drug delivery devices, which integrate sensors and apps for real-time adherence tracking and remote monitoring in chronic conditions like diabetes and asthma, face multifaceted market challenges that hinder their widespread adoption. High development and manufacturing costs driven by advanced sensors, R&D, precision assembly, and supply chain issues elevate prices above traditional devices, compounded by ongoing cloud fees, tariffs, and payer reimbursement gaps. Further, stringent regulatory hurdles for drug-device combinations and global compliance delays, clash with rapid tech evolution and limit AI innovations. In addition, cybersecurity and data privacy risks hamper user trust and restrict data sharing, while usage barriers like usability issues persist.
Connected Drug Delivery Devices Market: Key Insights
The report delves into the current state of the connected drug delivery devices market and identifies potential growth opportunities within industry. Some key findings from the report include:
- The current market landscape features the presence of more than 80 manufacturers that offer various connected drug delivery devices; of these, majority are headquartered in North America.
- Nearly 75% of the connected / smart drug delivery devices are currently commercially available; notably, most of these devices are intended for the administration of medications via subcutaneous route.

- At present, over 30 innovative add-on sensors are available / under development for integration with conventional drug delivery devices; numerous add-on sensor manufacturers are start-ups based in Europe.
- More than 55% of the add-on sensors are designed for drug delivery via inhalation; of these, over 85% of the sensors can support Bluetooth connectivity.
- The majority of clinical trials registered in this domain till date have evaluated the safety and efficacy of various connected drug delivery devices; most of these studies have been conducted across multiple sites in the US.
- The growing interest of stakeholders in this market is reflected from the rising partnerships established among various stakeholders in the recent past; in fact, close to 70% of the deals were inked in the last three years.
- Investors are increasingly channelizing capital into connected drug delivery devices market for the development of advanced devices which are accelerating industry advancements.
- The development process of a connected / smart drug delivery device involves various steps, ranging from planning, designing, engineering, process validation, manufacturing and assembly to commercialization.
- The pioneer-migrator-settler map in blue ocean strategy analysis suggests that several connected drug delivery devices manufacturers focused on enhancing their products are likely to emerge as pioneers in the coming years.

- Owing to the ability of connected drug delivery devices to provide precise dosing and remote patient monitoring, the market is anticipated to witness a notable growth (CAGR of 10.2%) over the next decade.

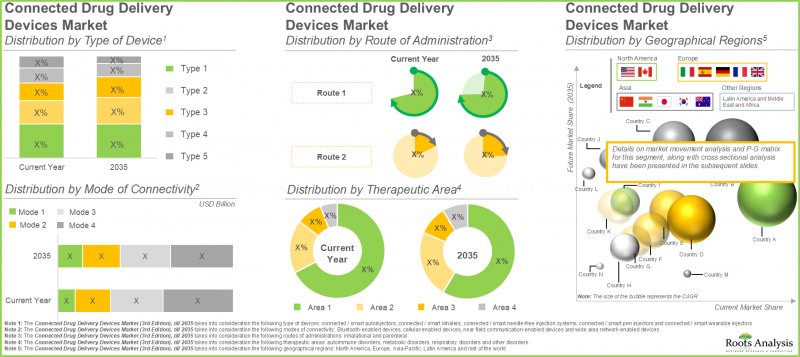
- The connected drug delivery devices market in North America is expected to capture maximum share by 2035; further, majority of the devices manufactured for drug delivery are intended for metabolic disorders.
- Driven by the growing adoption of smart and connected drug delivery solutions in the US, the connected drug delivery devices market is poised to grow at a higher CAGR over the forecast period.
- Given the surge in demand for advanced drug delivery devices and the rising prevalence of chronic diseases, the connected drug delivery devices market is anticipated to rise steadily.
Connected Drug Delivery Devices Market
The market sizing and opportunity analysis has been segmented across the following parameters:
By Type of Device
- Connected / Smart Wearable Injectors
- Connected / Smart Inhalers
- Connected / Smart Pen Injectors
- Connected / Smart Autoinjectors
- Connected / Smart Needle-free Injection Systems
By Mode of Connectivity
- Bluetooth-enabled Devices
- Near Field Communication-enabled Devices
- Cellular-enabled Devices
- Wide Area Network-enabled Devices
By Route of Administration
- Parenteral
- Inhalational
By Therapeutic Area
- Metabolic Disorders
- Respiratory Disorders
- Autoimmune Disorders
- Other Disorders
By Geographical Regions
- North America
- US
- Canada
- Europe
- UK
- Germany
- France
- Italy
- Spain
- Rest of Europe
- Asia-Pacific
- China
- India
- Japan
- Rest of Asia-Pacific
Connected Drug Delivery Devices Market: Key Segments
Connected / Smart Wearable Injectors Account for the Largest Market Share
In the current year, the connected / smart wearable injectors holds the largest market share, and this trend is expected to persist in the coming years. This is due to the increasing prevalence of chronic diseases and the ongoing initiatives of stakeholders to incorporate enhanced features into their connected drug delivery devices. These improvements enhance monitoring, strengthen patient compliance, and allow data exchange with healthcare professionals.
Bluetooth-Enabled Connected / Smart Devices Hold the Highest Share in the Future
At present, Bluetooth-enabled connected / smart devices are expected to capture a larger share of the connected drug delivery devices market. Moreover, this segment is expected to experience substantial growth during the forecasted period. This is because Bluetooth connectivity is simple to set up, widely accessible on all smartphones, and operates without requiring any additional devices.
Parenteral Segment Holds the Highest Connected Drug Delivery Devices Market Share in the Current Year
According to our estimates, in the current year, the parenteral segment is likely to hold a larger market share. This is because it is the favored route for administering biologics and high-value treatments for chronic conditions. Further, the inhalation segment is expected to experience a higher CAGR over the forecast period, owing to the ease, versatility, and painless nature of drug delivery through this method, along with its ability for prolonged localized therapeutic effect.
North America Dominates the Market by Securing Highest Share
According to our estimates, North America is expected to dominate the connected drug delivery devices market share this year. Further, the Asia-Pacific market is expected to expand at a comparatively higher rate till 2035. The growth is driven by a rising number of biopharmaceutical start-ups, significant research and development efforts for innovative drug delivery systems, and an increase in device approvals for various disease therapies in this area.
Primary Research Overview
Discussions with multiple stakeholders in this domain influenced the opinions and insights presented in this study. The market report includes transcripts of the following other third-party discussions:
- Senior Director, Global Strategic Marketing, Medical, Large Company, US
- Co-Founder and Chief Executive Officer, Small Company, US
- Chief Executive Officer, Small Company, US
- Chief Executive Officer and President, Mid-sized Company, US
- Chief Executive Officer, Small Company, Italy
- Vice President and Chief Scientist, Large Company, Israel
- Director of Global Communications, and Former Vice President of Strategic Partnerships and Business Development, Mid-sized Company, US
- Former Chief Executive Officer, Small Company, US
- Former Director of Front-end Innovation and Head of Connected Health, Large Company, US
- Former Vice President, Marketing and Alliance Management, Mid-sized Company, US
- Advisor of Strategy and Business Development, Small Company, US
Example Players in Connected Drug Delivery Devices Market
- Bigfoot Biomedical
- BIOCORP
- Eitan Medical
- E3D Elcam Drug Delivery Device
- EoFlow
- Gerresheimer
- Insulet
- Medtronic
- Medtrum
- Nemera
- Phillips-Medisize
- Roche
- Sonceboz
- SOOIL Development
- Tandem Diabetes Care
- West Pharmaceutical Services
- Ypsomed
Connected Drug Delivery Devices Market: Research Coverage
- Market Sizing and Opportunity Analysis: The report features an in-depth analysis of the connected drug delivery devices market, focusing on key market segments, including [A] type of device [B] mode of connectivity, [C] route of administration, [D] therapeutic area, and [E] geographical regions.
- Connected / Smart Drug Delivery Devices Market Landscape: A detailed overview of the overall market landscape of integrated connected drug delivery devices, along with information on several relevant parameters, such as [A] stage of development, [B] launch year of device, [C] type of integrated device, [D] route of administration, [E] target indication, therapeutic area, [F] key features, [G] mode of connectivity, [H] availability of companion mobile application and availability of data storage / cloud platform, [I] year of establishment, [J] company size, [K] location of headquarters and [H] most active players.
- Add-On Sensors Market Landscape: : A detailed overview overall market landscape of add-on sensors market landscape, along with information on several relevant parameters, such as [A] stage of development, [B] launch year of device, [C] compatible device, [D] route of administration, [E] target indication, [F] therapeutic area, [G] target indication, [H] key features, [I] mode of connectivity, [J] availability of companion mobile application and [K] availability of data storage / cloud platform, [L] year of establishment, [M] company size, [N] location of headquarters and [O] most active players.
- Product Competitiveness Analysis: An insightful analysis of connected drug delivery devices and add-on sensors based on the [A] manufacturer strength and [B] portfolio strength.
- Company Profiles: In-depth profiles of leading players manufacturing connected drug delivery devices in North America, Europe and Asia-Pacific based on [A] year of establishment, [B] location of headquarters, [C] product portfolio, [D] recent developments and [E] an informed future outlook.
- Clinical Trial Analysis: An analysis of completed and ongoing clinical trials related to connected drug delivery devices, based on several relevant parameters, such as [A] trial registration year, [B] enrolled patient population, [C] trial status, [D] type of sponsor / collaborator, [E] study design [F] and geography.
- Partnerships and Collaborations: An analysis of partnerships and collaborations inked between stakeholders, based on several relevant parameters, such as [A] year of partnership, [B] type of partnership, [C] class of device, [D] type of integrated device, [E] most active players.
- Funding and Investment Analysis: An analysis of funding and investment signed in the domain based on several relevant parameters, such as [A] year of funding, [B] type of funding, [C] amount invested, [D] geography, [E] and most active players.
- Value Chain Analysis: An in-depth value chain analysis featuring a discussion on various steps involved in the development of connected / smart drug delivery devices, including research and development (R&D), product manufacturing and assembly, product distribution, marketing and sales, and post-market surveillance, along with the information on cost requirements across each of the aforementioned stages.
- Regulatory Landscape: An informative summary of various guidelines established and issued by major regulatory bodies for the approval of connected drug delivery devices, across different countries / geographical regions.
- Case Studies: Detailed case studies on two most common indications (namely asthma and diabetes), featuring a brief description of the disease, its epidemiology, economic burden (imposed due to non-adherence to prescribed treatments), and a summary of the evolution of drug delivery devices and other strategies focused on improving therapy adherence among patients suffering from the indication.
- Blue Ocean Analysis: A detailed analysis of the current and future market based on blue ocean strategy, covering a strategic plan / guide for emerging connected drug delivery device companies to help unlock an uncontested market, featuring strategic tools that can help them to shift towards a blue ocean in order to gain a competitive edge in the market.
- SWOT Analysis: A detailed discussion on industry affiliated trends, opportunities and challenges, under a SWOT analysis, which are likely to impact the evolution of connected drug delivery devices market.
Key Questions Answered in this Report
- Which are the leading companies in connected drug delivery devices market?
- Which region dominates the connected drug delivery devices market?
- What are the key trends observed in the connected drug delivery devices market?
- What factors are likely to influence the evolution of this market?
- What are the primary challenges faced by connected drug delivery device developers?
- What is the current and future market size?
- What is the CAGR of this market?
- How is the current and future market opportunity likely to be distributed across key market segments?
Reasons to Buy this Report
- The report provides a comprehensive market analysis, offering detailed revenue projections of the overall market and its specific sub-segments. This information is valuable to both established market leaders and emerging entrants.
- The report offers stakeholders a comprehensive overview of the market, including key drivers, barriers, opportunities, and challenges. This information empowers stakeholders to stay abreast of market trends and make data-driven decisions to capitalize on growth prospects.
- The report can aid businesses in identifying future opportunities in any sector. It also helps in understanding if those opportunities are worth pursuing.
- The report helps in identifying customer demand by understanding the needs, preferences, and behavior of the target audience in order to tailor products or services effectively.
- The report equips new entrants with requisite information regarding a particular market to help them build successful business strategies.
- The report allows for more effective communication with the audience and in building strong business relations.
Additional Benefits
- Complementary PPT Insights Pack
- Complimentary Excel Data Packs for all Analytical Modules in the Report
- 15% Free Content Customization
- Detailed Report Walkthrough Session with Research Team
- Free Updated report if the report is 6-12 months old or older
TABLE OF CONTENTS
1. PREFACE
- 1.1. Introduction
- 1.2. Market Share Insights
- 1.3. Key Market Insights
- 1.4. Report Coverage
- 1.5. Key Questions Answered
- 1.6. Chapter Outlines
2. RESEARCH METHODOLOGY
- 2.1. Chapter Overview
- 2.2. Research Assumptions
- 2.2.1. Market Landscape and Market Trends
- 2.2.2. Market Forecast and Opportunity Analysis
- 2.2.3. Comparative Analysis
- 2.3. Database Building
- 2.3.1. Data Collection
- 2.3.2. Data Validation
- 2.3.3. Data Analysis
- 2.4. Project Methodology
- 2.4.1. Secondary Research
- 2.4.1.1. Annual Reports
- 2.4.1.2. Academic Research Papers
- 2.4.1.3. Company Websites
- 2.4.1.4. Investor Presentations
- 2.4.1.5. Regulatory Filings
- 2.4.1.6. White Papers
- 2.4.1.7. Industry Publications
- 2.4.1.8. Conferences and Seminars
- 2.4.1.9. Government Portals
- 2.4.1.10. Media and Press Releases
- 2.4.1.11. Newsletters
- 2.4.1.12. Industry Databases
- 2.4.1.13. Roots Proprietary Databases
- 2.4.1.14. Paid Databases and Sources
- 2.4.1.15. Social Media Portals
- 2.4.1.16. Other Secondary Sources
- 2.4.2. Primary Research
- 2.4.2.1. Types of Primary Research
- 2.4.2.1.1. Qualitative Research
- 2.4.2.1.2. Quantitative Research
- 2.4.2.1.3. Hybrid Approach
- 2.4.2.2. Advantages of Primary Research
- 2.4.2.3. Techniques for Primary Research
- 2.4.2.3.1. Interviews
- 2.4.2.3.2. Surveys
- 2.4.2.3.3. Focus Groups
- 2.4.2.3.4. Observational Research
- 2.4.2.3.5. Social Media Interactions
- 2.4.2.4. Key Opinion Leaders Considered in Primary Research
- 2.4.2.4.1. Company Executives (CXOs)
- 2.4.2.4.2. Board of Directors
- 2.4.2.4.3. Company Presidents and Vice Presidents
- 2.4.2.4.4. Research and Development Heads
- 2.4.2.4.5. Technical Experts
- 2.4.2.4.6. Subject Matter Experts
- 2.4.2.4.7. Scientists
- 2.4.2.4.8. Doctors and Other Healthcare Providers
- 2.4.2.5. Ethics and Integrity
- 2.4.2.5.1. Research Ethics
- 2.4.2.5.2. Data Integrity
- 2.4.2.1. Types of Primary Research
- 2.4.3. Analytical Tools and Databases
- 2.4.1. Secondary Research
- 2.5. Robust Quality Control
3. MARKET DYNAMICS
- 3.1. Chapter Overview
- 3.2. Forecast Methodology
- 3.2.1. Top-down Approach
- 3.2.2. Bottom-up Approach
- 3.2.3. Hybrid Approach
- 3.3. Market Assessment Framework
- 3.3.1. Total Addressable Market (TAM)
- 3.3.2. Serviceable Addressable Market (SAM)
- 3.3.3. Serviceable Obtainable Market (SOM)
- 3.3.4. Currently Acquired Market (CAM)
- 3.4. Forecasting Tools and Techniques
- 3.4.1. Qualitative Forecasting
- 3.4.2. Correlation
- 3.4.3. Regression
- 3.4.4. Extrapolation
- 3.4.5. Convergence
- 3.4.6. Sensitivity Analysis
- 3.4.7. Scenario Planning
- 3.4.8. Data Visualization
- 3.4.9. Time Series Analysis
- 3.4.10. Forecast Error Analysis
- 3.5. Key Considerations
- 3.5.1. Demographics
- 3.5.2. Government Regulations
- 3.5.3. Reimbursement Scenarios
- 3.5.4. Market Access
- 3.5.5. Supply Chain
- 3.5.6. Industry Consolidation
- 3.5.7. Pandemic / Unforeseen Disruptions Impact
- 3.6. Limitations
4. MACRO-ECONOMIC INDICATORS
- 4.1. Chapter Overview
- 4.2. Market Dynamics
- 4.2.1. Time Period
- 4.2.1.1. Historical Trends
- 4.2.1.2. Current and Forecasted Estimates
- 4.2.2. Currency Coverage
- 4.2.2.1. Major Currencies Affecting the Market
- 4.2.2.2. Factors Affecting Currency Fluctuations on the Industry
- 4.2.2.3. Impact of Currency Fluctuations on the Industry
- 4.2.3. Foreign Currency Exchange Rate
- 4.2.3.1. Impact of Foreign Exchange Rate Volatility on the Market
- 4.2.3.2. Strategies for Mitigating Foreign Exchange Risk
- 4.2.4. Recession
- 4.2.4.1. Assessment of Current Economic Conditions and Potential Impact on the Market
- 4.2.4.2. Historical Analysis of Past Recessions and Lessons Learnt
- 4.2.5. Inflation
- 4.2.5.1. Measurement and Analysis of Inflationary Pressures in the Economy
- 4.2.5.2. Potential Impact of Inflation on the Market Evolution
- 4.2.6. Interest Rates
- 4.2.6.1. Interest Rates and Their Impact on the Market
- 4.2.6.2. Strategies for Managing Interest Rate Risk
- 4.2.7. Commodity Flow Analysis
- 4.2.7.1. Type of Commodity
- 4.2.7.2. Origins and Destinations
- 4.2.7.3. Values and Weights
- 4.2.7.4. Modes of Transportation
- 4.2.8. Global Trade Dynamics
- 4.2.8.1. Import Scenario
- 4.2.8.2. Export Scenario
- 4.2.8.3. Trade Policies
- 4.2.8.4. Strategies for Mitigating the Risks Associated with Trade Barriers
- 4.2.8.5. Impact of Trade Barriers on the Market
- 4.2.9. War Impact Analysis
- 4.2.9.1. Russian-Ukraine War
- 4.2.9.2. Israel-Hamas War
- 4.2.10. COVID Impact / Related Factors
- 4.2.10.1. Global Economic Impact
- 4.2.10.2. Industry-specific Impact
- 4.2.10.3. Government Response and Stimulus Measures
- 4.2.10.4. Future Outlook and Adaptation Strategies
- 4.2.11. Other Indicators
- 4.2.11.1. Fiscal Policy
- 4.2.11.2. Consumer Spending
- 4.2.11.3. Gross Domestic Product
- 4.2.11.4. Employment
- 4.2.11.5. Taxes
- 4.2.11.6. Stock Market Performance
- 4.2.11.7. Cross Border Dynamics
- 4.2.1. Time Period
- 4.3. Conclusion
5. EXECUTIVE SUMMARY
6. INTRODUCTION
- 6.1. Chapter Overview
- 6.2. Medication Adherence
- 6.2.1. Digital Tools for Improving Medication Adherence
- 6.3. Overview of Connected / Smart Drug Delivery Devices
- 6.4. Key Design Features and Development Process
- 6.5. Classification of Connected / Smart Drug Delivery Devices
- 6.5.1. Type of Device
- 6.5.1.1. Connected Inhalers
- 6.5.1.2. Connected Handheld Injectors
- 6.5.1.3. Smart Pills
- 6.5.1.4. Smart Implantable Devices
- 6.5.2. Type of Technology
- 6.5.1. Type of Device
- 6.6. Regulatory Guidelines for Connected / Smart Drug Delivery Devices
- 6.7. Advantages and Limitations of Connected / Smart Drug Delivery Devices
- 6.8. Impact of COVID-19 on Connected / Smart Drug Delivery Devices Domain
- 6.9. Future Perspective
7. MARKET LANDSCAPE: CONNECTED / SMART DRUG DELIVERY DEVICES
- 7.1. Chapter Overview
- 7.2. Connected / Smart Drug Delivery Devices: Overall Market Landscape
- 7.2.1. Analysis by Stage of Development
- 7.2.2. Analysis by Launch Year
- 7.2.3. Analysis by Type of Integrated Device
- 7.2.4. Analysis by Route of Administration
- 7.2.5. Analysis by Target Indication
- 7.2.6. Analysis by Therapeutic Area
- 7.2.7. Analysis by Key Features
- 7.2.8. Analysis by Mode of Connectivity
- 7.2.9. Analysis by Availability of Companion Mobile Application
- 7.2.10. Analysis by Availability of Data Storage / Cloud Platform
- 7.3. Connected / Smart Drug Delivery Device Manufacturers: Overall Market Landscape
- 7.3.1. Analysis by Year of Establishment
- 7.3.2. Analysis by Company Size
- 7.3.3. Analysis by Location of Headquarters
- 7.3.4. Analysis by Year of Establishment and Location of Headquarters
- 7.3.5. Analysis by Company Size and Location of Headquarters
- 7.3.6. Most Active Players: Analysis by Number of Connected / Smart Drug Delivery Devices Manufactured
8. MARKET LANDSCAPE: ADD-ON SENSORS
- 8.1. Chapter Overview
- 8.2. Add-on Sensors: Overall Market Landscape
- 8.2.1. Analysis by Stage of Development
- 8.2.2. Analysis by Launch Year
- 8.2.3. Analysis by Compatible Device
- 8.2.4. Analysis by Route of Administration
- 8.2.5. Analysis by Target Indication
- 8.2.6. Analysis by Therapeutic Area
- 8.2.7. Analysis by Key Features
- 8.2.8. Analysis by Mode of Connectivity
- 8.2.9. Analysis by Availability of Companion Mobile Application
- 8.2.10. Analysis by Availability of Data Storage / Cloud Platform
- 8.3. Add-on Sensor Manufacturers: Overall Market Landscape
- 8.3.1. Analysis by Year of Establishment
- 8.3.2. Analysis by Company Size
- 8.3.3. Analysis by Location of Headquarters
- 8.3.4. Analysis by Year of Establishment and Location of Headquarters
- 8.3.5. Analysis by Company Size and Location of Headquarters
- 8.3.6. Most Active Players: Analysis by Number of Add-on Sensors Manufactured
9. PRODUCT COMPETITIVENESS ANALYSIS
- 9.1. Chapter Overview
- 9.2. Assumptions and Key Parameters
- 9.3. Methodology
- 9.4. Connected Drug Delivery Devices and Add-on Sensors: Product Competitiveness Analysis
- 9.4.1. Connected / Smart Drug Delivery Devices
- 9.4.1.1. Smart Handheld Injectors (Peer Group 1a)
- 9.4.1.2. Smart Wearable Injectors (Peer Group 1b)
- 9.4.1.3. Other Integrated Devices (Peer Group 1c)
- 9.4.2. Add-on Sensors (Peer Group 2)
- 9.4.1. Connected / Smart Drug Delivery Devices
10. COMPANY PROFILES: CONNECTED / SMART DRUG DELIVERY DEVICE MANUFACTURERS BASED IN NORTH AMERICA
- 10.1. Chapter Overview
- 10.2. Leading Connected / Smart Drug Delivery Device Manufacturers
- 10.2.1. Medtronic
- 10.2.1.1. Company Overview
- 10.2.1.2. Financial Information
- 10.2.1.3. Connected / Smart Drug Delivery Devices Portfolio
- 10.2.1.4. Recent Developments and Future Outlook
- 10.2.2. Insulet
- 10.2.2.1. Company Overview
- 10.2.2.2. Financial Information
- 10.2.2.3. Connected / Smart Drug Delivery Devices Portfolio
- 10.2.2.4. Recent Developments and Future Outlook
- 10.2.3. Tandem Diabetes Care
- 10.2.3.1. Company Overview
- 10.2.3.2. Financial Information
- 10.2.3.3. Connected / Smart Drug Delivery Devices Portfolio
- 10.2.3.4. Recent Developments and Future Outlook
- 10.2.1. Medtronic
- 10.3. Other Prominent Connected / Smart Drug Delivery Device Manufacturers
- 10.3.1. Bigfoot Biomedical
- 10.3.1.1. Company Overview
- 10.3.1.2. Connected / Smart Drug Delivery Devices Portfolio
- 10.3.2. Phillips Medisize (a subsidiary of Molex)
- 10.3.2.1. Company Overview
- 10.3.2.2. Connected / Smart Drug Delivery Devices Portfolio
- 10.3.3. West Pharmaceutical Services
- 10.3.3.1. Company Overview
- 10.3.3.2. Connected / Smart Drug Delivery Devices Portfolio
- 10.3.1. Bigfoot Biomedical
11. COMPANY PROFILES: CONNECTED / SMART DRUG DELIVERY DEVICE MANUFACTURERS BASED IN EUROPE
- 11.1. Chapter Overview
- 11.2. Leading Connected / Smart Drug Delivery Device Manufacturers
- 11.2.1. BIOCORP
- 11.2.1.1. Company Overview
- 11.2.1.2. Financial Information
- 11.2.1.3. Connected / Smart Drug Delivery Devices Portfolio
- 11.2.1.4. Recent Developments and Future Outlook
- 11.2.2. Gerresheimer
- 11.2.2.1. Company Overview
- 11.2.2.2. Financial Information
- 11.2.2.3. Connected / Smart Drug Delivery Devices Portfolio
- 11.2.2.4. Recent Developments and Future Outlook
- 11.2.3. Roche
- 11.2.3.1. Company Overview
- 11.2.3.2. Financial Information
- 11.2.3.3. Connected / Smart Drug Delivery Devices Portfolio
- 11.2.3.4. Recent Developments and Future Outlook
- 11.2.4. Ypsomed
- 11.2.4.1. Company Overview
- 11.2.4.2. Financial Information
- 11.2.4.3. Connected / Smart Drug Delivery Devices Portfolio
- 11.2.4.4. Recent Developments and Future Outlook
- 11.2.1. BIOCORP
- 11.3. Other Prominent Connected / Smart Drug Delivery Device Manufacturers
- 11.3.1. Nemera
- 11.3.1.1. Company Overview
- 11.3.1.2. Connected / Smart Drug Delivery Devices Portfolio
- 11.3.2. Sonceboz
- 11.3.2.1. Company Overview
- 11.3.2.2. Connected / Smart Drug Delivery Devices Portfolio
- 11.3.1. Nemera
12. COMPANY PROFILES: CONNECTED / SMART DRUG DELIVERY DEVICE MANUFACTURERS BASED IN ASIA-PACIFIC AND REST OF THE WORLD
- 12.1. Chapter Overview
- 12.2. Leading Connected / Smart Drug Delivery Device Manufacturers
- 12.2.1. Medtrum
- 12.2.1.1. Company Overview
- 12.2.1.2. Connected / Smart Drug Delivery Devices Portfolio
- 12.2.1.3. Recent Developments and Future Outlook
- 12.2.2. SOOIL Development
- 12.2.2.1. Company Overview
- 12.2.2.2. Connected / Smart Drug Delivery Devices Portfolio
- 12.2.2.3. Recent Developments and Future Outlook
- 12.2.3. EOFlow
- 12.2.3.1. Company Overview
- 12.2.3.2. Financial Information
- 12.2.3.3. Connected / Smart Drug Delivery Devices Portfolio
- 12.2.3.4. Recent Developments and Future Outlook
- 12.2.1. Medtrum
- 12.3. Other Prominent Connected / Smart Drug Delivery Device Manufacturers
- 12.3.1. Eitan Medical
- 12.3.1.1. Company Overview
- 12.3.1.2. Connected / Smart Drug Delivery Devices Portfolio
- 12.3.2. E3D Elcam Drug Delivery Device
- 12.3.2.1. Company Overview
- 12.3.2.2. Connected / Smart Drug Delivery Devices Portfolio
- 12.3.1. Eitan Medical
13. CLINICAL TRIAL ANALYSIS
- 13.1. Chapter Overview
- 13.2. Scope and Methodology
- 13.3. Connected / Smart Drug Delivery Devices and Add-on Sensors: Clinical Trial Analysis
- 13.3.1. Analysis by Trial Registration Year
- 13.3.2. Analysis of Enrolled Patient Population by Trial Registration Year
- 13.3.3. Analysis by Trial Status
- 10.3.4. Analysis by Trial Registration Year and Trial Status
- 13.3.5. Analysis of Number of Enrolled Patients Population by Trial Status
- 13.3.6. Analysis by Target Patient Population
- 13.3.7. Analysis by Type of Sponsor / Collaborator
- 13.3.8. Analysis by Study Design
- 10.3.8.1. Analysis by Type of Allocation
- 10.3.8.2. Analysis by Type of Intervention Model
- 10.3.8.3. Analysis by Type of Masking
- 10.3.8.4. Analysis by Trial Purpose
- 13.3.9. Leading Players: Analysis by Number of Registered Trials
- 13.3.10. Leading Connected / Smart Drug Delivery Devices and Add-on Sensors: Analysis by Number of Registered Trials
- 13.3.11. Analysis by Geography
- 13.3.11.1. Analysis of Clinical Trials by Geography
- 13.3.11.2. Analysis of Number of Registered Trials by Trial Status and Geography
- 13.3.11.3. Analysis of Number of Enrolled Patient Population by Geography
14. PARTNERSHIPS AND COLLABORATIONS
- 14.1. Chapter Overview
- 14.2. Partnership Models
- 14.3. Connected / Smart Drug Delivery Devices: Partnerships and Collaborations
- 14.3.1. Analysis by Year of Partnership
- 14.3.2. Analysis by Type of Partnership
- 14.3.3. Analysis by Year and Type of Partnership
- 14.3.4. Analysis by Class of Device
- 14.3.5. Analysis by Type of Integrated Device
- 14.3.6. Most Active Players: Analysis by Number of Partnerships
- 14.3.7. Analysis by Geography
- 14.3.7.1. Intracontinental and Intercontinental Deals
- 14.3.7.2. Local and International Deals
15. FUNDING AND INVESTMENT ANALYSIS
- 15.1. Chapter Overview
- 15.2. Funding Models
- 15.3. Connected / Smart Drug Delivery Devices: Funding and Investments
- 15.3.1. Analysis by Year of Funding
- 15.3.2. Analysis by Type of Funding
- 15.3.3. Analysis by Year and Type of Funding
- 15.3.4. Analysis of Amount Invested by Year of Funding
- 15.3.5. Analysis of Amount Invested by Type of Funding
- 15.3.6. Analysis by Geography
- 15.3.7. Most Active Players: Analysis by Number of Funding Instances
- 15.3.8. Most Active Players: Analysis by Amount Raised
16. VALUE CHAIN ANALYSIS
- 16.1. Chapter Overview
- 16.2. Connected / Smart Drug Delivery Devices: Value Chain
- 16.3. Cost Distribution across the Value Chain
- 16.3.1. Concept Development and Proof of Concept
- 16.3.2. Research and Product Development
- 16.3.3. Conduct of Clinical Trials
- 16.3.4. Regulatory Approvals
- 16.3.5. Manufacturing and Product Assembly
- 16.3.6. Application Development
- 16.3.7. Marketing and Sales
17. REGULATORY LANDSCAPE FOR CONNECTED / SMART DRUG DELIVERY DEVICES
- 17.1. Chapter Overview
- 17.2. Regulatory Approval of Combination Products in North America
- 17.2.1. Regulatory Approval of Combination Products in the US
- 17.2.1.1. Overview
- 17.2.1.2. Historical Background
- 17.2.1.3. Role of Regulatory Bodies in Product Approval
- 17.2.2. Regulatory Approval of Combination Products in Canada
- 17.2.2.1. Overview
- 17.2.2.2. Role of Regulatory Bodies in Product Approval
- 17.2.3. Regulatory Approval of Combination Products in Mexico
- 17.2.3.1. Overview
- 17.2.3.2. Role of Regulatory Bodies in Product Approval
- 17.2.1. Regulatory Approval of Combination Products in the US
- 17.3. Regulatory Approval of Combination Products in Europe
- 17.3.1. Regulatory Approval of Combination Products in EU5 Countries
- 17.3.1.1. Overview
- 17.3.1.2. Role of Regulatory Bodies in Product Approval
- 17.3.1. Regulatory Approval of Combination Products in EU5 Countries
- 17.4. Regulatory Approval of Combination Products in Asia-Pacific
- 17.4.1. Regulatory Approval of Combination Products in Japan
- 17.4.1.1. Overview
- 17.4.1.2. Role of Regulatory Bodies in Product Approval
- 17.4.2. Regulatory Approval of Combination Products in China
- 17.4.2.1. Overview
- 17.4.2.2. Role of Regulatory Bodies in Product Approval
- 17.4.3. Regulatory Approval of Combination Products in India
- 17.4.3.1. Overview
- 17.4.3.2. Role of Regulatory Bodies in Product Approval
- 17.4.4. Regulatory Approval of Combination Products in South Korea
- 17.4.4.1. Overview
- 17.4.4.2. Role of Regulatory Bodies in Product Approval
- 17.4.5. Regulatory Approval of Combination Products in Australia
- 17.4.5.1. Overview
- 17.4.5.2. Role of Regulatory Bodies in Product Approval
- 17.4.1. Regulatory Approval of Combination Products in Japan
- 17.5. Regulatory Approval of Combination Products in Middle East and North Africa
- 17.5.1. Regulatory Approval of Combination Products in Saudi Arabia
- 17.5.1.1. Overview
- 17.5.1.2. Role of Regulatory Bodies in Product Approval
- 17.5.2. Regulatory Approval of Combination Products in United Arab Emirates
- 17.5.2.1. Overview
- 17.5.2.2. Role of Regulatory Bodies in Product Approval
- 17.5.1. Regulatory Approval of Combination Products in Saudi Arabia
- 17.6. Regulatory Approval of Combination Products in Latin America
- 17.6.1. Regulatory Approval of Combination Products in Brazil
- 17.6.1.1. Overview
- 17.6.1.2. Role of Regulatory Bodies in Product Approval
- 17.6.2. Regulatory Approval of Combination Products in Argentina
- 17.6.2.1. Overview
- 17.6.2.2. Role of Regulatory Bodies in Product Approval
- 17.6.1. Regulatory Approval of Combination Products in Brazil
- 17.7. Connected Devices: Other Measures for Testing Safety, Effectiveness and Performance
- 17.8. Ensuring Security of Data Captured
- 17.9. Concluding Remarks
18. CASE STUDIES: APPLICATION OF CONNECTED DRUG DELIVERY DEVICES TO IMPROVE MEDICATION ADHERENCE
- 18.1. Chapter Overview
- 18.2. Case Study I: Diabetes
- 18.2.1. Disease Overview
- 18.2.2. Key Statistics
- 18.2.3. Economic Burden due to Non-Adherence
- 18.2.4. Evolution of Connected / Smart Drug Delivery Devices for Improving Adherence
- 18.3. Case Study II: Asthma
- 18.3.1. Disease Overview
- 18.3.2. Key Statistics
- 18.3.3. Economic Burden due to Non-Adherence
- 18.3.4. Evolution of Connected / Smart Drug Delivery Devices for Improving Adherence
19. SWOT ANALYSIS
- 19.1. Chapter Overview
- 19.2. Strengths
- 19.2.1. Ability to Minimize / Eliminate Medication Errors
- 19.2.2. Economic Advantages
- 19.2.3. Technological Advancements
- 19.3. Weaknesses
- 19.3.1. Cybersecurity
- 19.3.2. Stringent Regulatory Approvals
- 19.3.3. High Developmental Costs
- 19.4. Opportunities
- 19.4.1. Growing Prevalence of Chronic Disorders
- 19.4.2. Increasing Adoption of Digital Health Technologies
- 19.4.3. Rising Partnership and Investment Activity
- 19.5. Threats
- 19.5.1. Material Compatibility Issues
- 19.5.2. Availability of Alternative Drug Delivery Devices
- 19.5.3. Concern Related to Product Recalls in Past
- 19.6. Comparison of SWOT Factors
20. BLUE OCEAN STRATEGY
- 20.1. Overview of Blue Ocean Strategy
- 20.1.1. Red Oceans
- 20.1.2. Blue Oceans
- 20.1.3. Comparison of Red Ocean Strategy and Blue Ocean Strategy
- 20.1.4. Connected Drug Delivery Devices: Blue Ocean Strategy and Shift Tools
- 20.1.4.1. Strategy Canvas
- 20.1.4.2. Pioneer-Migrator-Settler (PMS) Map
21. GLOBAL CONNECTED DRUG DELIVERY DEVICES MARKET
- 21.1. Chapter Overview
- 21.2. Assumptions and Methodology
- 21.3. Global Connected Drug Delivery Devices Market: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 21.3.1. Scenario Analysis
- 21.3.1.1. Conservative Scenario
- 21.3.1.2. Optimistic Scenario
- 21.3.1. Scenario Analysis
- 21.4. Key Market Segmentations
22. CONNECTED DRUG DELIVERY DEVICES MARKET, BY TYPE OF DEVICE
- 22.1. Chapter Overview
- 22.2. Assumptions and Methodology
- 22.3. Connected Drug Delivery Devices Market: Distribution by Type of Device
- 22.3.1. Connected Drug Delivery Devices Market for Connected / Smart Wearable Injectors: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 22.3.2. Connected Drug Delivery Devices Market for Connected / Smart Inhalers: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 22.3.3. Connected Drug Delivery Devices Market for Connected / Smart Pen Injectors: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 22.3.4. Connected Drug Delivery Devices Market for Connected / Smart Autoinjectors: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 22.3.5. Connected Drug Delivery Devices Market for Connected / Smart Needle-free Injection Systems: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 22.4. Data Triangulation and Validation
23. CONNECTED DRUG DELIVERY DEVICES MARKET, BY MODE OF CONNECTIVITY
- 23.1. Chapter Overview
- 23.2. Assumptions and Methodology
- 23.3. Connected Drug Delivery Devices Market: Distribution by Mode of Connectivity
- 23.3.1. Connected Drug Delivery Devices Market for Bluetooth-enabled Devices: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 23.3.2. Connected Drug Delivery Devices Market for Near-field Communication-enabled Devices: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 23.3.3. Connected Drug Delivery Devices Market for Cellular-enabled Devices: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 23.3.4. Connected Drug Delivery Devices Market for Wide Area Network-enabled Devices: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 23.4. Data Triangulation and Validation
24. CONNECTED DRUG DELIVERY DEVICES MARKET, BY ROUTE OF ADMINISTRATION
- 24.1. Chapter Overview
- 24.2. Assumptions and Methodology
- 24.3. Connected Drug Delivery Devices Market: Distribution by Route of Administration
- 24.3.1. Connected Drug Delivery Devices Market for Parenteral Administration: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 24.3.2. Connected Drug Delivery Devices Market for Inhalational Administration: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 24.4. Data Triangulation and Validation
25. CONNECTED DRUG DELIVERY DEVICES MARKET, BY THERAPEUTIC AREA
- 25.1. Chapter Overview
- 25.2. Assumptions and Methodology
- 25.3. Connected Drug Delivery Devices Market: Distribution by Therapeutic Area
- 25.3.1. Connected Drug Delivery Devices Market for Metabolic Disorders: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 25.3.2. Connected Drug Delivery Devices Market for Respiratory Disorders: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 25.3.3. Connected Drug Delivery Devices Market for Autoimmune Disorders: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 25.3.4. Connected Drug Delivery Devices Market for Other Disorders: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 25.4. Data Triangulation and Validation
26. CONNECTED DRUG DELIVERY DEVICES MARKET, BY GEOGRAPHICAL REGIONS
- 26.1. Chapter Overview
- 26.2. Assumptions and Methodology
- 26.3. Connected Drug Delivery Devices Market: Distribution by Geographical Regions
- 26.3.1. Connected Drug Delivery Devices Market in North America: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 26.3.1.1. Connected Drug Delivery Devices Market in the US: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 26.3.1.2. Connected Drug Delivery Devices Market in Canada: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 26.3.2. Connected Drug Delivery Devices Market in Europe: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 26.3.2.1. Connected Drug Delivery Devices Market in the UK: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 26.3.2.2. Connected Drug Delivery Devices Market in Germany: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 26.3.2.3. Connected Drug Delivery Devices Market in France: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 26.3.2.4. Connected Drug Delivery Devices Market in Italy: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 26.3.2.5. Connected Drug Delivery Devices Market in Spain: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 26.3.2.6. Connected Drug Delivery Devices Market in Rest of Europe: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 26.3.3. Connected Drug Delivery Devices Market in Asia-Pacific: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 26.3.3.1. Connected Drug Delivery Devices Market in China: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 26.3.3.2. Connected Drug Delivery Devices Market in India: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 26.3.3.3. Connected Drug Delivery Devices Market in Japan: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 26.3.3.4. Connected Drug Delivery Devices Market in Rest of Asia-Pacific: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 26.3.4. Connected Drug Delivery Devices Market in Middle East and North Africa: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 26.3.4.1. Connected Drug Delivery Devices Market in Egypt: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 26.3.4.2. Connected Drug Delivery Devices Market in Israel: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 26.3.4.3. Connected Drug Delivery Devices Market in Saudi Arabia: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 26.3.4.4. Connected Drug Delivery Devices Market in Rest of Middle East and North Africa: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 26.3.5. Connected Drug Delivery Devices Market in Latin America and Rest of the World: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 26.3.5.1. Connected Drug Delivery Devices Market in Brazil: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 26.3.5.2. Connected Drug Delivery Devices Market in Argentina: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 26.3.5.3. Connected Drug Delivery Devices Market in Other Countries: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 26.3.1. Connected Drug Delivery Devices Market in North America: Historical Trends (since 2020) and Forecasted Estimates (till 2035)
- 26.4. Data Triangulation and Validation
27. CONNECTED DRUG DELIVERY DEVICES MARKET, BY LEADING PLAYERS
28. CONCLUDING REMARKS
29. EXECUTIVE INSIGHTS
30. APPENDIX 1: TABULATED DATA
31. APPENDIX 2: LIST OF COMPANIES AND ORGANIZATIONS
32. APPENDIX 3: PARTNERSHIPS AND COLLABORATIONS, 2015-2020
List of Tables
- Table 7.1 Connected / Smart Drug Delivery Devices: Information on Manufacturer, Stage of Development, Launch Year, Type of Integrated Device and Route of Administration
- Table 7.2 Connected / Smart Drug Delivery Devices: Information on Therapeutic Area, Target Indication and Key Features
- Table 7.3 Connected / Smart Drug Delivery Devices: Information on Mode of Connectivity, Availability of Companion Mobile Application, Compatible Operating System and Availability of Data Storage / Cloud Platform
- Table 7.4 Connected / Smart Drug Delivery Devices: Information on Regulatory Approvals, Geographical Reach and Compatible Drug
- Table 7.5 Connected / Smart Drug Delivery Device Manufacturers: Information on Year of Establishment, Company Size and Location of Headquarters
- Table 8.1 Add-on Sensors: Information on Manufacturer, Stage of Development, Launch Year, Type of Integrated Device and Route of Administration
- Table 8.2 Add-on Sensors: Information on Therapeutic Area, Target Indication and Key Features
- Table 8.3 Add-on Sensors: Information on Mode of Connectivity, Availability of Companion Mobile Application, and Availability of Data Storage / Cloud Platform
- Table 8.4 Add-on Sensors: Information on Regulatory Approval, Geographical Reach and Drug Delivered
- Table 8.5 Add-on Sensor Manufacturers: Information on Year of Establishment, Company Size and Location of Headquarters
- Table 9.1 Product Competitiveness Analysis: Connected / Smart Drug Delivery Devices
- Table 10.1 Connected / Smart Drug Delivery Device Manufacturers in North America: List of Companies Profiled
- Table 10.2 Medtronic: Company Overview
- Table 10.3 Medtronic: Connected / Smart Drug Delivery Devices Portfolio
- Table 10.4 Medtronic: Recent Developments and Future Outlook
- Table 10.5 Insulet: Company Overview
- Table 10.6 Insulet: Connected / Smart Drug Delivery Devices Portfolio
- Table 10.7 Insulet: Recent Developments and Future Outlook
- Table 10.8 Tandem Diabetes Care: Company Overview
- Table 10.9 Tandem Diabetes Care: Connected / Smart Drug Delivery Devices Portfolio
- Table 10.10 Tandem Diabetes Care: Recent Developments and Future Outlook
- Table 10.11 Bigfoot Biomedical: Company Overview
- Table 10.12 Bigfoot Biomedical: Connected / Smart Drug Delivery Devices Portfolio
- Table 10.13 Phillips-Medsize (a subsidiary of Molex): Company Overview
- Table 10.14 Phillips-Medsize (a subsidiary of Molex): Connected / Smart Drug Delivery Devices Portfolio
- Table 10.15 West Pharmaceutical Services: Company Overview
- Table 10.16 West Pharmaceutical Services: Connected / Smart Drug Delivery Devices Portfolio
- Table 11.1 Connected / Smart Drug Delivery Device Manufacturers in Europe: List of Companies Profiled
- Table 11.2 BIOCORP: Company Overview
- Table 11.3 BIOCORP: Connected / Smart Drug Delivery Devices Portfolio
- Table 11.4 BIOCORP: Recent Developments and Future Outlook
- Table 11.5 Gerresheimer: Company Overview
- Table 11.6 Gerresheimer: Connected / Smart Drug Delivery Devices Portfolio
- Table 11.7 Gerresheimer: Recent Developments and Future Outlook
- Table 11.8 Roche: Company Overview
- Table 11.9 Roche: Connected / Smart Drug Delivery Devices Portfolio
- Table 11.10 Roche: Recent Developments and Future Outlook
- Table 11.11 Ypsomed: Company Overview
- Table 11.12 Ypsomed: Connected / Smart Drug Delivery Devices Portfolio
- Table 11.13 Ypsomed: Recent Developments and Future Outlook
- Table 11.14 Nemera: Company Overview
- Table 11.15 Nemera: Connected / Smart Drug Delivery Devices Portfolio
- Table 11.16 Sonceboz: Company Overview
- Table 11.17 Sonceboz: Connected / Smart Drug Delivery Devices Portfolio
- Table 12.1 Connected / Smart Drug Delivery Device Manufacturers in Asia-Pacific and Rest of the World: List of Companies Profiled
- Table 12.2 Medtrum: Company Overview
- Table 12.3 Medtrum: Connected / Smart Drug Delivery Devices Portfolio
- Table 12.4 SOOIL Development: Company Overview
- Table 12.5 SOOIL Development: Connected / Smart Drug Delivery Devices Portfolio
- Table 12.6 SOOIL Development: Recent Developments and Future Outlook
- Table 12.7 EOFlow: Company Overview
- Table 12.8 EOFlow: Connected / Smart Drug Delivery Devices Portfolio
- Table 12.9 EOFlow: Recent Developments and Future Outlook
- Table 12.10 Eitan Medical: Company Overview
- Table 12.11 Eitan Medical: Connected / Smart Drug Delivery Devices Portfolio
- Table 12.12 E3D Elcam Drug Delivery Device: Company Overview
- Table 12.13 E3D Elcam Drug Delivery Device: Connected / Smart Drug Delivery Devices Portfolio
- Table 14.1 Connected / Smart Drug Delivery Devices: List of Partnerships and Collaborations, since 2021
- Table 15.1 Connected / Smart Drug Delivery Devices: List of Funding and Investments, since 2021
- Table 17.1 FDA Centers for Drug and Device Approval
- Table 17.2 Regulatory Review Timelines for Combination Products in the US
- Table 17.3 Regulatory Bodies in EU5 Countries
- Table 17.4 SFDA: Regulatory Review Timeline
- Table 27.1 Connected Drug Delivery Devices Market: Leading Industry Players
- Table 27.1 Company A: Company Overview
- Table 27.2 Company B: Company Overview
- Table 27.3 Company C: Company Overview
- Table 27.4 Company D: Company Overview
- Table 27.5 Company E: Company Overview
- Table 27.6 Company F: Company Overview
- Table 27.7 Company G: Company Overview
- Table 27.8 Company H: Company Overview
- Table 27.9 Company I: Company Overview
- Table 27.10 Company J: Company Overview
- Table 27.11 Company K: Company Overview
- Table 28.1 Connected / Smart Drug Delivery Devices: Distribution by Stage of Development
- Table 28.2 Connected / Smart Drug Delivery Devices: Distribution by Launch Year
- Table 28.3 Connected / Smart Drug Delivery Devices: Distribution by Type of Integrated Device
- Table 28.4 Connected / Smart Drug Delivery Devices: Distribution by Route of Administration
- Table 28.5 Connected / Smart Drug Delivery Devices: Distribution by Therapeutic Area
- Table 28.6 Connected / Smart Drug Delivery Devices: Distribution by Target Indication
- Table 28.7 Connected / Smart Drug Delivery Devices: Distribution by Key Features
- Table 28.8 Connected / Smart Drug Delivery Devices: Distribution by Mode of Connectivity
- Table 28.9 Connected / Smart Drug Delivery Devices: Distribution by Availability of Companion Mobile Application
- Table 28.10 Connected / Smart Drug Delivery Devices: Distribution by Availability of Data Storage / Cloud Platform
- Table 28.11 Connected / Smart Drug Delivery Device Manufacturers: Distribution by Year of Establishment
- Table 28.12 Connected / Smart Drug Delivery Device Manufacturers: Distribution by Company Size
- Table 28.13 Connected / Smart Drug Delivery Device Manufacturers: Distribution by Location of Headquarters
- Table 28.14 Connected / Smart Drug Delivery Device Manufacturers: Distribution by Year of Establishment and Location of Headquarters (Region)
- Table 28.15 Connected / Smart Drug Delivery Device Manufacturers: Distribution by Company Size and Location of Headquarters (Region)
- Table 28.16 Most Active Players: Distribution by Number of Connected / Smart Drug Delivery Devices Manufactured
- Table 28.17 Add-on Sensors: Distribution by Stage of Development
- Table 28.18 Add-on Sensors: Distribution by Launch Year
- Table 28.19 Add-on Sensors: Distribution by Compatible Device
- Table 28.20 Add-on Sensors: Distribution by Route of Administration
- Table 28.21 Add-on Sensors: Distribution by Target Indication
- Table 28.22 Add-on Sensors: Distribution by Therapeutic Area
- Table 28.23 Add-on Sensors: Distribution by Key Features
- Table 28.24 Add-on Sensors: Distribution by Mode of Connectivity
- Table 28.25 Add-on Sensors: Distribution by Availability of Companion Mobile Application
- Table 28.26 Add-on Sensors: Distribution by Availability of Data Storage / Cloud Platform
- Table 28.27 Add-on Sensor Manufacturers: Distribution by Year of Establishment
- Table 28.28 Add-on Sensor Manufacturers: Distribution by Company Size
- Table 28.29 Add-on Sensor Manufacturers: Distribution by Location of Headquarters
- Table 28.30 Add-on Sensor Manufacturers: Distribution by Year of Establishment and Location of Headquarters
- Table 28.31 Add-on Sensor Manufacturers: Distribution by Company Size and Location of Headquarters
- Table 28.32 Most Active Players: Distribution by Number of Add-on Sensors Manufactured
- Table 28.33 Medtronic: Business Segment-wise Revenues and Consolidated Financial Details (USD Billion)
- Table 28.34 Insulet: Business Segment-wise Revenues and Consolidated Financial Details (USD Million)
- Table 28.35 Tandem Diabetes Care: Consolidated Financial Details (USD Million)
- Table 28.36 Gerresheimer: Business Segment-wise Revenues and Consolidated Financial Details (EUR Million)
- Table 28.37 Roche: Business Segment-wise Revenues and Consolidated Financial Details (CHF Billion)
- Table 28.38 Ypsomed: Business Segment-wise Revenues and Consolidated Financial Details (CHF Million)
- Table 28.39 Clinical Trial Analysis: Distribution by Trial Registration Year, since 2021
- Table 28.40 Clinical Trial Analysis: Year-wise Trend of Patients Enrolled by Trial Registration Year, since 2021
- Table 28.41 Clinical Trial Analysis: Distribution by Trial Status
- Table 28.42 Clinical Trial Analysis: Distribution by Enrolled Patient Population by Trial Registration Year
- Table 28.43 Clinical Trial Analysis: Distribution by Trial Registration Year and Trial Status
- Table 28.44 Clinical Trial Analysis: Distribution of Number of Enrolled Patient Population by Trial Status
- Table 28.45 Clinical Trial Analysis: Distribution by Target Patient Population
- Table 28.46 Clinical Trial Analysis: Distribution by Type of Sponsor / Collaborator
- Table 28.47 Clinical Trial Analysis: Distribution by Study Design
- Table 28.48 Clinical Trial Analysis: Distribution by Type of Allocation
- Table 28.49 Clinical Trial Analysis: Distribution by Type of Intervention Model
- Table 28.50 Clinical Trial Analysis: Distribution by Type of Masking
- Table 28.51 Clinical Trial Analysis: Distribution by Trial Purpose
- Table 28.52 Leading Players: Distribution by Number of Registered Trials
- Table 28.53 Leading Connected / Smart Drug Delivery Devices and Add-On Sensors: Distribution by Number of Registered Trials
- Table 28.54 Clinical Trial Analysis: Distribution of Clinical Trials by Geography
- Table 28.55 Clinical Trial Analysis: Distribution of Number of Registered Trials by Trial Status and Geography
- Table 28.56 Clinical Trial Analysis: Distribution of Number of Enrolled Patients Population by Geography
- Table 28.57 Partnerships and Collaborations: Cumulative Year-wise Trend, since 2021
- Table 28.58 Partnerships and Collaborations: Distribution by Type of Partnership
- Table 28.59 Partnerships and Collaborations: Distribution by Year and Type of Partnership, since 2021
- Table 28.60 Partnerships and Collaborations: Distribution by Class of Device
- Table 28.61 Partnerships and Collaborations: Distribution by Type of Integrated Device
- Table 28.62 Partnerships and Collaborations: Distribution by Compatible Device
- Table 28.63 Most Active Players: Distribution by Number of Partnerships
- Table 28.64 Partnerships and Collaborations: Intercontinental and Intracontinental Deals
- Table 28.65 Partnerships and Collaborations: Local and International Deals
- Table 28.66 Funding and Investment Analysis: Cumulative Year-wise Trend, since 2021
- Table 28.67 Funding and Investment Analysis: Distribution by Amount Invested, since 2021 (USD Million)
- Table 28.68 Funding and Investment Analysis: Distribution of Funding Instances by Type of Funding
- Table 28.69 Funding and Investments: Distribution by Amount Invested by Type of Funding (USD Million)
- Table 28.70 Distribution of Funding Instances by Year and Type of Funding, since 2021
- Table 28.71 Funding and Investments: Distribution of Funding Instances by Geography
- Table 28.72 Most Active Players: Distribution by Number of Funding Instances
- Table 28.73 Most Active Players: Distribution by Amount Raised (USD Million)
- Table 28.74 Funding and Investments Summary, since 2021 (USD Million)
- Table 28.75 Global Connected Drug Delivery Devices Demand, Historical Trends, since 2020 (Million Units Sold)
- Table 28.76 Global Connected Drug Delivery Devices Demand, till 2035 (Million Units Sold)
- Table 28.77 Global Connected Drug Delivery Devices Market, Historical Trends, Since 2020 (USD Million)
- Table 28.78 Global Connected Drug Delivery Devices Market, till 2035 (USD Million)
- Table 28.79 Connected Drug Delivery Devices Market: Distribution by Type of Device (USD Million)
- Table 28.80 Connected Drug Delivery Devices Demand for Connected / Smart Wearable Injectors, Historical Trends, since 2020 (Million Units Sold)
- Table 28.81 Connected Drug Delivery Devices Demand for Connected / Smart Wearable Injectors, till 2035 (Million Units Sold)
- Table 28.82 Connected Drug Delivery Devices Market for Connected / Smart Wearable Injectors, Historical Trends, Since 2020 (USD Million)
- Table 28.83 Connected Drug Delivery Devices Market for Connected / Smart Wearable Injectors, till 2035 (USD Million)
- Table 28.84 Connected Drug Delivery Devices Demand for Connected / Smart Inhalers, Historical Trends, since 2020 (Million Units Sold)
- Table 28.85 Connected Drug Delivery Devices Demand for Connected / Smart Inhalers, till 2035 (Million Units Sold)
- Table 28.86 Connected Drug Delivery Devices Market for Connected / Smart Inhalers, Historical Trends, since 2020 (USD Million)
- Table 28.87 Connected Drug Delivery Devices Market for Connected / Smart Inhalers, till 2035 (USD Million)
- Table 28.88 Connected Drug Delivery Devices Demand for Connected / Smart Pen Injectors, Historical Trends, since 2020 (Million Units Sold)
- Table 28.89 Connected Drug Delivery Devices Demand for Connected / Smart Pen Injectors, till 2035 (Million Units Sold)
- Table 28.90 Connected Drug Delivery Devices Market for Connected / Smart Pen Injectors, Historical Trends, since 2020 (USD Million)
- Table 28.91 Connected Drug Delivery Devices Market for Connected / Smart Pen Injectors, till 2035 (USD Million)
- Table 28.92 Connected Drug Delivery Devices Demand for Connected / Smart Autoinjectors, Historical Trends, since 2020 (Million Units Sold)
- Table 28.93 Connected Drug Delivery Devices Demand for Connected / Smart Autoinjectors, till 2035 (Million Units Sold)
- Table 28.94 Connected Drug Delivery Devices Market for Connected / Smart Autoinjectors, Historical Trends, since 2020 (USD Million)
- Table 28.95 Connected Drug Delivery Devices Market for Connected / Smart Autoinjectors, till 2035 (USD Million)
- Table 28.96 Connected Drug Delivery Devices Demand for Connected / Smart Needle-free Injection Systems, till 2035 (Million Units Sold)
- Table 28.97 Connected Drug Delivery Devices Demand for Connected / Smart Needle-free Injection Systems, till 2035 (USD Million)
- Table 28.98 Connected Drug Delivery Devices Market: Distribution by Mode of Connectivity, 2020, 2024 and 2035 (USD Million)
- Table 28.99 Connected Drug Delivery Devices Demand for Bluetooth-enabled Devices, Historical Trends, since 2020 (Million Units Sold)
- Table 28.100 Connected Drug Delivery Devices Demand for Bluetooth-enabled Devices, till 2035 (Million Units Sold)
- Table 28.101 Connected Drug Delivery Devices Market for Bluetooth-enabled Devices, Historical Trends, since 2020 (USD Million)
- Table 28.102 Connected Drug Delivery Devices Market for Bluetooth-enabled Devices, till 2035 (USD Million)
- Table 28.103 Connected Drug Delivery Devices Demand for Near-field Communication-enabled Devices, Historical Trends, since 2020 (Million Units Sold)
- Table 28.104 Connected Drug Delivery Devices Demand for Near-field Communication-enabled Devices, till 2035 (Million Units Sold)
- Table 28.105 Connected Drug Delivery Devices Market for Near-field Communication-enabled Devices, Historical Trends, since 2020 (USD Million)
- Table 28.106 Connected Drug Delivery Devices Market for Near-field Communication-enabled Devices, till 2035 (USD Million)
- Table 28.107 Connected Drug Delivery Devices Demand for Cellular-enabled Devices, Historical Trends, since 2020 (Million Units Sold)
- Table 28.108 Connected Drug Delivery Devices Demand for Cellular-enabled Devices, till 2035 (Million Units Sold)
- Table 28.109 Connected Drug Delivery Devices Market for Cellular-enabled Devices, Historical Trends, since 2020 (USD Million)
- Table 28.110 Connected Drug Delivery Devices Market for Cellular-enabled Devices, till 2035 (USD Million)
- Table 28.111 Connected Drug Delivery Devices Demand for Wide Area Network-enabled Devices, Historical Trends, since 2020 (Million Units Sold)
- Table 28.112 Connected Drug Delivery Devices Demand for Wide Area Network-enabled Devices, till 2035 (Million Units Sold)
- Table 28.113 Connected Drug Delivery Devices Market for Wide Area Network-enabled Devices, Historical Trends, since 2020 (USD Million)
- Table 28.114 Connected Drug Delivery Devices Market for Wide Area Network-enabled Devices, till 2035 (USD Million)
- Table 28.115 Connected Drug Delivery Devices Market: Distribution by Route of Administration, 2020, 2024 and 2035 (USD Million)
- Table 28.116 Connected Drug Delivery Demand for Parenteral Administration, Historical Trends, since 2020 (Million Units Sold)
- Table 28.117 Connected Drug Delivery Devices Demand for Parenteral Administration, till 2035 (Million Units Sold)
- Table 28.118 Connected Drug Delivery Devices Market for Parenteral Administration, Historical Trends, since 2020 (USD Million)
- Table 28.119 Connected Drug Delivery Devices Market for Parenteral Administration, till 2035 (USD Million)
- Table 28.120 Connected Drug Delivery Devices Demand for Inhalational Administration, Historical Trends, since 2020 (Million Units Sold)
- Table 28.121 Connected Drug Delivery Devices Demand for Inhalational Administration, till 2035 (Million Units Sold)
- Table 28.122 Connected Drug Delivery Devices Market for Inhalational Administration, Since 2020 (USD Million)
- Table 28.123 Connected Drug Delivery Devices Market for Inhalational Administration, till 2035 (USD Million)
- Table 28.124 Connected Drug Delivery Devices Market: Distribution by Therapeutic Area, 2020, 2024 and 2035 (USD Million)
- Table 28.125 Connected Drug Delivery Devices Demand for Metabolic Disorders, Historical Trends, since 2020 (Million Units Sold)
- Table 28.126 Connected Drug Delivery Devices Demand for Metabolic Disorders, till 2035 (Million Units Sold)
- Table 28.127 Connected Drug Delivery Devices Market for Metabolic Disorders, Historical Trends, since 2020 (USD Million)
- Table 28.128 Connected Drug Delivery Devices Market for Metabolic Disorders, till 2035 (USD Million)
- Table 28.129 Connected Drug Delivery Devices Demand for Respiratory Disorders, Historical Trends, since 2020 (Million Units Sold)
- Table 28.130 Connected Drug Delivery Devices Demand for Respiratory Disorders, till 2035 (Million Units Sold)
- Table 28.131 Connected Drug Delivery Devices Market for Respiratory Disorders, Historical Trends, since 2020 (USD Million)
- Table 28.132 Connected Drug Delivery Devices Market for Respiratory Disorders, till 2035 (USD Million)
- Table 28.133 Connected Drug Delivery Devices Demand for Autoimmune Disorders, Historical Trends, since 2020 (Million Units Sold)
- Table 28.134 Connected Drug Delivery Devices Demand for Autoimmune Disorders, till 2035 (Million Units Sold)
- Table 28.135 Connected Drug Delivery Devices Market for Autoimmune Disorders, Historical Trends, since 2020 (USD Million)
- Table 28.136 Connected Drug Delivery Devices Market for Autoimmune Disorders, till 2035 (USD Million)
- Table 28.137 Connected Drug Delivery Devices Demand for Other Disorders, Historical Trends, since 2020 (Million Units Sold)
- Table 28.138 Connected Drug Delivery Devices Demand for Other Disorders, till 2035 (Million Units Sold)
- Table 28.139 Connected Drug Delivery Devices Market for Other Disorders, Historical Trends, since 2020 (USD Million)
- Table 28.140 Connected Drug Delivery Devices Market for Other Disorders, till 2035 (USD Million)
- Table 28.141 Connected Drug Delivery Devices Market: Distribution by Geographical Regions, 2020, 2024 and 2035 (USD Million)
- Table 28.142 Connected Drug Delivery Devices Demand in North America, Historical Trends, since 2020 (Million Units Sold)
- Table 28.143 Connected Drug Delivery Devices Demand in North America, till 2035 (Million Units Sold)
- Table 28.144 Connected Drug Delivery Devices Market in North America, Historical Trends, since 2020 (USD Million)
- Table 28.145 Connected Drug Delivery Devices Market in North America, till 2035 (USD Million)
- Table 28.146 Connected Drug Delivery Devices Market in the US, Historical Trends, Since 2020 (USD Million)
- Table 28.147 Connected Drug Delivery Devices Market in the US, till 2035 (USD Million)
- Table 28.148 Connected Drug Delivery Devices Market in Canada, Historical Trends, since 2020 (USD Million)
- Table 28.149 Connected Drug Delivery Devices Market in Canada, till 2035 (USD Million)
- Table 28.150 Connected Drug Delivery Devices Demand in Europe, Historical Trends, since 2020 (Million Units Sold)
- Table 28.151 Connected Drug Delivery Devices Demand in Europe, till 2035 (Million Units Sold)
- Table 28.152 Connected Drug Delivery Devices Market in Europe, Historical Trends, since 2020 (USD Million)
- Table 28.153 Connected Drug Delivery Devices Market in Europe, till 2035 (USD Million)
- Table 28.154 Connected Drug Delivery Devices Market in the UK, Historical Trends, since 2020 (USD Million)
- Table 28.155 Connected Drug Delivery Devices Market in the UK, till 2035 (USD Million)
- Table 28.156 Connected Drug Delivery Devices Market in Germany, Historical Trends, since 2020 (USD Million)
- Table 28.157 Connected Drug Delivery Devices Market in Germany, till 2035 (USD Million)
- Table 28.158 Connected Drug Delivery Devices Market in France, Historical Trends, Since 2020 (USD Million)
- Table 28.159 Connected Drug Delivery Devices Market in France, till 2035 (USD Million)
- Table 28.160 Connected Drug Delivery Devices Market in Italy, Historical Trends, since 2020 (USD Million)
- Table 28.161 Connected Drug Delivery Devices Market in Italy, till 2035 (USD Million)
- Table 28.162 Connected Drug Delivery Devices Market in Spain, Historical Trends, since 2020 (USD Million)
- Table 28.163 Connected Drug Delivery Devices Market in Spain, till 2035 (USD Million)
- Table 28.164 Connected Drug Delivery Devices Market in Rest of Europe, Historical Trends, since 2020 (USD Million)
- Table 28.165 Connected Drug Delivery Devices Market in Rest of Europe, till 2035 (USD Million)
- Table 28.166 Connected Drug Delivery Devices Demand in Asia-Pacific, Historical Trends, since 2020 (Million Units Sold)
- Table 28.167 Connected Drug Delivery Devices Demand in Asia-Pacific, till 2035 (Million Units Sold)
- Table 28.168 Connected Drug Delivery Devices Market in Asia-Pacific, Historical Trends, since 2020 (USD Million)
- Table 28.169 Connected Drug Delivery Devices Market in Asia-Pacific, till 2035 (USD Million)
- Table 28.170 Connected Drug Delivery Devices Market in China, Historical Trends, since 2020 (USD Million)
- Table 28.171 Connected Drug Delivery Devices Market in China, till 2035 (USD Million)
- Table 28.172 Connected Drug Delivery Devices Market in India, Historical Trends, Since 2020 (USD Million)
- Table 28.173 Connected Drug Delivery Devices Market in India, till 2035 (USD Million)
- Table 28.174 Connected Drug Delivery Devices Market in Japan, Historical Trends, Since 2020 (USD Million)
- Table 28.175 Connected Drug Delivery Devices Market in Japan, till 2035 (USD Million)
- Table 28.176 Connected Drug Delivery Devices Market in Rest of the Asia-Pacific, Historical Trends, since 2020 (USD Million)
- Table 28.177 Connected Drug Delivery Devices Market in Rest of the Asia-Pacific, till 2035 (USD Million)
- Table 28.178 Connected Drug Delivery Devices Demand in Middle East and North Africa, Historical Trends, since 2020 (Million Units Sold)
- Table 28.179 Connected Drug Delivery Devices Demand in Middle East and North Africa, till 2035 (Million Units Sold)
- Table 28.180 Connected Drug Delivery Devices Market in Middle East and North Africa, Historical Trends, Since 2020 (USD Million)
- Table 28.181 Connected Drug Delivery Devices Market in Middle East and North Africa, till 2035 (USD Million)
- Table 28.182 Connected Drug Delivery Devices Market in Egypt, Historical Trends, since 2020 (USD Million)
- Table 28.183 Connected Drug Delivery Devices Market in Egypt, till 2035 (USD Million)
- Table 28.184 Connected Drug Delivery Devices Market in Israel, Historical Trends, since 2020 (USD Million)
- Table 28.185 Connected Drug Delivery Devices Market in Israel, till 2035 (USD Million)
- Table 28.186 Connected Drug Delivery Devices Market in Saudi Arabia, Historical Trends, since 2020 (USD Million)
- Table 28.187 Connected Drug Delivery Devices Market in Saudi Arabia, till 2035 (USD Million)
- Table 28.188 Connected Drug Delivery Devices Market in Rest of the Middle East and North Africa, Historical Trends, since 2020 (USD Million)
- Table 28.189 Connected Drug Delivery Devices Market in Rest of the Middle East and North Africa, till 2035 (USD Million)
- Table 28.190 Connected Drug Delivery Devices Demand in Latin America and Rest of the World, Historical Trends, since 2020 (Million Units Sold)
- Table 28.191 Connected Drug Delivery Devices Demand in Latin America and Rest of the World, till 2035 (Million Units Sold)
- Table 28.192 Connected Drug Delivery Devices Market in Latin America and Rest of the World, Historical Trends, since 2020 (USD Million)
- Table 28.193 Connected Drug Delivery Devices Market in Latin America and Rest of the World, till 2035 (USD Million)
- Table 28.194 Connected Drug Delivery Devices Market in Brazil, Historical Trends, since 2020 (USD Million)
- Table 28.195 Connected Drug Delivery Devices Market in Brazil, till 2035 (USD Million)
- Table 28.196 Connected Drug Delivery Devices Market in Argentina, Historical Trends, since 2020 (USD Million)
- Table 28.197 Connected Drug Delivery Devices Market in Argentina, till 2035 (USD Million)
- Table 28.198 Connected Drug Delivery Devices Market in Rest of the World, Historical Trends, since 2020 (USD Million)
- Table 28.199 Connected Drug Delivery Devices Market in Rest of the World, till 2035 (USD Million)
List of Figures
- Figure 2.1 Research Methodology: Project Methodology
- Figure 2.2 Research Methodology: Data Sources for Secondary Research
- Figure 3.1 Market Dynamics: Forecast Methodology
- Figure 3.2 Market Dynamics: Key Market Segmentation
- Figure 3.3 Market Dynamics: Robust Quality Control
- Figure 4.1 Lessons Learnt from Past Recessions
- Figure 5.1 Executive Summary: Connected / Smart Drug Delivery Devices Market Landscape
- Figure 5.2 Executive Summary: Add-on Sensors Market Landscape
- Figure 5.3 Executive Summary: Market Trends
- Figure 5.4 Executive Summary: Market Forecast and Opportunity Analysis
- Figure 6.1 Different Components of IoT Ecosystem
- Figure 6.2 Framework of Connected / Smart Drug Delivery Devices
- Figure 6.3 Key Considerations for Designing and Developing a Connected / Smart Drug Delivery Device
- Figure 6.4 Stack Model for the Development of Connected Drug Delivery Devices
- Figure 6.5 Type of Technology Used for Connected / Smart Drug Delivery Device
- Figure 6.6 Advantages of Connected / Smart Drug Delivery Devices
- Figure 7.1 Connected / Smart Drug Delivery Devices: Distribution by Stage of Development
- Figure 7.2 Connected / Smart Drug Delivery Devices: Distribution by Launch Year
- Figure 7.3 Connected / Smart Drug Delivery Devices: Distribution by Type of Integrated Device
- Figure 7.4 Connected / Smart Drug Delivery Devices: Distribution by Route of Administration
- Figure 7.5 Connected / Smart Drug Delivery Devices: Distribution by Target Indication
- Figure 7.6 Connected / Smart Drug Delivery Devices: Distribution by Therapeutic Area
- Figure 7.7 Connected / Smart Drug Delivery Devices: Distribution by Key Features
- Figure 7.8 Connected / Smart Drug Delivery Devices: Distribution by Mode of Connectivity
- Figure 7.9 Connected / Smart Drug Delivery Devices: Distribution by Availability of Companion Mobile Application
- Figure 7.10 Connected / Smart Drug Delivery Devices: Distribution by Availability of Data Storage / Cloud Platform
- Figure 7.11 Connected / Smart Drug Delivery Device Manufacturers: Distribution by Year of Establishment
- Figure 7.12 Connected / Smart Drug Delivery Device Manufacturers: Distribution by Company Size
- Figure 7.13 Connected / Smart Drug Delivery Device Manufacturers: Distribution by Location of Headquarters
- Figure 7.14 Connected / Smart Drug Delivery Device Manufacturers: Distribution by Year of Establishment and Location of Headquarters
- Figure 7.15 Connected / Smart Drug Delivery Device Manufacturers: Distribution by Company Size and Location of Headquarters
- Figure 7.16 Most Active Players: Distribution by Number of Connected / Smart Drug Delivery Devices Manufactured
- Figure 8.1 Add-on Sensors: Distribution by Stage of Development
- Figure 8.2 Add-on Sensors: Distribution by Launch Year
- Figure 8.3 Add-on Sensors: Distribution by Compatible Device
- Figure 8.4 Add-on Sensors: Distribution by Route of Administration
- Figure 8.5 Add-on Sensors: Distribution by Target Indication
- Figure 8.6 Add-on Sensors: Distribution by Therapeutic Area
- Figure 8.7 Add-on Sensors: Distribution by Key Features
- Figure 8.8 Add-on Sensors: Distribution by Mode of Connectivity
- Figure 8.9 Add-on Sensors: Distribution by Availability of Companion Mobile Application
- Figure 8.10 Add-on Sensors: Distribution by Availability of Data Storage / Cloud Platform
- Figure 8.11 Add-on Sensor Manufacturers: Distribution by Year of Establishment
- Figure 8.12 Add-on Sensor Manufacturers: Distribution by Company Size
- Figure 8.13 Add-on Sensor Manufacturers: Distribution by Location of Headquarters
- Figure 8.14 Add-on Sensor Manufacturers: Distribution by Year of Establishment and Location of Headquarters
- Figure 8.15 Add-on Sensor Manufacturers: Distribution by Company Size and Location of Headquarters
- Figure 8.16 Most Active Players: Distribution by Number of Add-on Sensors Manufactured
- Figure 9.1 Product Competitiveness Analysis: Smart Handheld Injectors (Peer Group 1a)
- Figure 9.2 Product Competitiveness Analysis: Smart Wearable Injectors (Peer Group 1b)
- Figure 9.3 Product Competitiveness Analysis: Other Integrated Devices (Peer Group 1c)
- Figure 9.4 Product Competitiveness Analysis: Add-on Sensors (Peer Group 2)
- Figure 10.1 Medtronic: Business Segment-wise Revenues and Consolidated Financial Details (USD Billion)
- Figure 10.2 Insulet: Business Segment-wise Revenues and Consolidated Financial Details (USD Millon)
- Figure 10.3 Tandem Diabetes Care: Consolidated Financial Details (USD Million)
- Figure 11.1 Gerresheimer: Business Segment-wise Revenues and Consolidated Financial Details (EUR Million)
- Figure 11.2 Roche: Business Segment-wise Revenues and Consolidated Financial Details (CHF Billion)
- Figure 11.3 Ypsomed: Business Segment-wise Revenues and Consolidated Financial Details (CHF Million)
- Figure 13.1 Clinical Trial Analysis: Cumulative Year-wise Trend, since 2021
- Figure 13.2 Clinical Trial Analysis: Year-wise Trend of Enrolled Patient Population by Trial Registration Year, since 2021
- Figure 13.3 Clinical Trial Analysis: Distribution by Trial Status
- Figure 13.4 Clinical Trial Analysis: Distribution by Trial Registration Year and Trial Status
- Figure 13.5 Clinical Trial Analysis: Distribution of Number of Enrolled Patient Population by Trial Status
- Figure 13.6 Clinical Trial Analysis: Distribution by Target Patient Population
- Figure 13.7 Clinical Trial Analysis: Distribution by Type of Sponsor / Collaborator
- Figure 13.8 Clinical Trial Analysis: Distribution by Study Design
- Figure 13.9 Clinical Trial Analysis: Distribution by Type of Allocation
- Figure 13.10 Clinical Trial Analysis: Distribution by Type of Intervention Model
- Figure 13.11 Clinical Trial Analysis: Distribution by Type of Masking
- Figure 13.12 Clinical Trial Analysis: Distribution by Trial Purpose
- Figure 13.13 Leading Players: Distribution by Number of Registered Trials
- Figure 13.14 Leading Connected / Smart Drug Delivery Devices and Add-On Sensors: Distribution by Number of Registered Trials
- Figure 13.15 Clinical Trial Analysis: Distribution of Clinical Trials by Geography
- Figure 13.16 Clinical Trial Analysis: Distribution of Number of Registered Trials by Trial Status and Geography
- Figure 13.17 Clinical Trial Analysis: Distribution of Number of Enrolled Patients Population by Geography
- Figure 14.1 Partnerships and Collaborations: Cumulative Year-wise Trend, since 2021
- Figure 14.2 Partnerships and Collaborations: Distribution by Type of Partnership
- Figure 14.3 Partnerships and Collaborations: Distribution by Year and Type of Partnership, since 2021
- Figure 14.4 Partnerships and Collaborations: Distribution by Class of Device
- Figure 14.5 Partnerships and Collaborations: Distribution by Type of Integrated Device
- Figure 14.6 Partnerships and Collaborations: Distribution by Compatible Device
- Figure 14.7 Most Active Players: Distribution by Number of Partnerships
- Figure 14.8 Partnerships and Collaborations: Intracontinental and Intercontinental Deals
- Figure 14.9 Partnerships and Collaborations: Local and International Deals
- Figure 15.1 Funding and Investments: Cumulative Year-wise Trend, since 2021
- Figure 15.2 Funding and Investments: Distribution by Amount Invested, since 2021 (USD Million)
- Figure 15.3 Funding and Investments: Distribution of Funding Instances by Type of Funding
- Figure 15.4 Funding and Investments: Distribution of Amount Invested by Type of Funding (USD Million)
- Figure 15.5 Funding and Investments: Distribution of Funding Instances by Year and Type of Funding, since 2021
- Figure 15.6 Funding and Investments: Distribution of Funding Instances by Geography
- Figure 15.7 Most Active Players: Distribution by Number of Funding Instances
- Figure 15.8 Most Active Players: Distribution by Amount Raised (USD Million)
- Figure 15.9 Summary of Funding and Investments, since 2021 (USD Million)
- Figure 16.1 Connected / Smart Drug Delivery Devices: Value Chain
- Figure 16.2 Cost Distribution Across Value Chain
- Figure 16.3 Costs Associated with Concept Development and Proof of Concept
- Figure 16.4 Costs Associated with Research and Product Development
- Figure 16.5 Costs Associated with Clinical Trials
- Figure 16.6 Costs Associated with Regulatory Approvals
- Figure 16.7 Costs Associated with Manufacturing and Product Assembly
- Figure 16.8 Costs Associated with Application Development
- Figure 16.9 Costs Associated with Marketing and Sales
- Figure 17.1 Global Regulations related to Smart Inhalers, Smart Injectors and Smart Wearable Injectors
- Figure 17.2 USFDA: Approval Pathway for Combination Products
- Figure 17.3 Health Canada: Approval Pathway for Combination Products
- Figure 17.4 NMPA: Approval Pathway for Combination Products
- Figure 17.5 Connected / Smart Drug Delivery Devices: Major Steps for Managing Privacy
- Figure 18.1 Diabetes: Evolution of Drug Delivery / Connected Devices
- Figure 18.2 Impact of Drug Delivery / Connected Devices in Improving Drug Adherence in Diabetic Patients: Highlights on Twitter
- Figure 18.3 Asthma: Evolution of Drug Delivery / Connected Devices
- Figure 18.4 Impact of Drug Delivery / Connected Devices in Improving Drug Adherence in Asthmatic Patients: Highlights on Twitter
- Figure 19.1 Connected Drug Delivery Devices Market: SWOT Analysis
- Figure 19.2 Comparison of SWOT Factors: Harvey Ball Analysis
- Figure 20.1 Blue Ocean Strategy: Strategy Canvas
- Figure 20.2 Blue Ocean Strategy: Pioneer-Migrator (PMS) Map
- Figure 21.1 Global Connected Drug Delivery Devices Market: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (Million Units Sold, USD Million)
- Figure 21.2 Global Connected Drug Delivery Devices Market, till 2035: Conservative Scenario (Million Units Sold, USD Million)
- Figure 21.3 Global Connected Drug Delivery Devices Market, till 2035: Optimistic Scenario (Million Units Sold, USD Million)
- Figure 22.1 Connected Drug Delivery Devices Market: Distribution by Type of Device (USD Million)
- Figure 22.2 Connected Drug Delivery Devices Market for Connected / Smart Wearable Injectors: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (Million Units Sold, USD Million)
- Figure 22.3 Connected Drug Delivery Devices Market for Connected / Smart Inhalers: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (Million Units Sold, USD Million)
- Figure 22.4 Connected Drug Delivery Devices Market for Connected / Smart Pen Injectors: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (Million Units Sold, USD Million)
- Figure 22.5 Connected Drug Delivery Devices Market for Connected / Smart Autoinjectors: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (USD Million, Million Units Sold)
- Figure 22.6 Connected Drug Delivery Devices Market for Connected / Smart Needle-free Injection Systems: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (USD Million, Million Units Sold)
- Figure 23.1 Connected Drug Delivery Devices Market: Distribution by Mode of Connectivity (USD Million)
- Figure 23.2 Connected Drug Delivery Devices Market for Bluetooth-enabled Devices: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (Million Units Sold, USD Million)
- Figure 23.3 Connected Drug Delivery Devices Market for Near-field Communication-enabled Devices: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (Million Units Sold, USD Million)
- Figure 23.4 Connected Drug Delivery Devices Market for Cellular-enabled Devices: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (Million Units Sold, USD Million)
- Figure 23.5 Connected Drug Delivery Devices Market for Wide Area Network-enabled Devices: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (Million Units Sold, USD Million)
- Figure 24.1 Connected Drug Delivery Devices Market: Distribution by Route of Administration (USD Million)
- Figure 24.2 Connected Drug Delivery Devices Market for Parenteral Administration: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (Million Units Sold, USD Million)
- Figure 24.3 Connected Drug Delivery Devices Market for Inhalational Administration: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (Million Units Sold, USD Million)
- Figure 25.1 Connected Drug Delivery Devices Market: Distribution by Therapeutic Area (USD Million)
- Figure 25.2 Connected Drug Delivery Devices Market for Metabolic Disorders: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (Million Units Sold, USD Million)
- Figure 25.3 Connected Drug Delivery Devices Market for Respiratory Disorders: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (Million Units Sold, USD Million)
- Figure 25.4 Connected Drug Delivery Devices Market for Autoimmune Disorders: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (Million Units Sold, USD Million)
- Figure 25.5 Connected Drug Delivery Devices Market for Other Disorders: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (Million Units Sold, USD Million)
- Figure 26.1 Connected Drug Delivery Devices Market: Distribution by Geographical Regions (USD Million)
- Figure 26.2 Connected Drug Delivery Devices Market in North America: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (Million Units Sold, USD Million)
- Figure 26.3 Connected Drug Delivery Devices Market in the US: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (USD Million)
- Figure 26.4 Connected Drug Delivery Devices Market in Canada: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (USD Million)
- Figure 26.5 Connected Drug Delivery Devices Market in Europe: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (Million Units Sold, USD Million)
- Figure 26.6 Connected Drug Delivery Devices Market in the UK: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (USD Million)
- Figure 26.7 Connected Drug Delivery Devices Market in Germany: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (USD Million)
- Figure 26.8 Connected Drug Delivery Devices Market in France: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (USD Million)
- Figure 26.9 Connected Drug Delivery Devices Market in Italy: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (USD Million)
- Figure 26.10 Connected Drug Delivery Devices Market in Spain: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (USD Million)
- Figure 26.11 Connected Drug Delivery Devices Market in Rest of Europe: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (USD Million)
- Figure 26.12 Connected Drug Delivery Devices Market in Asia-Pacific: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (Million Units Sold, USD Million)
- Figure 26.13 Connected Drug Delivery Devices Market in China: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (USD Million)
- Figure 26.14 Connected Drug Delivery Devices Market in India: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (USD Million)
- Figure 26.15 Connected Drug Delivery Devices Market in Japan: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (USD Million)
- Figure 26.16 Connected Drug Delivery Devices Market in Rest of Asia-Pacific: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (USD Million)
- Figure 26.17 Connected Drug Delivery Devices Market in Middle East and North Africa: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (Million Units Sold, USD Million)
- Figure 26.18 Connected Drug Delivery Devices Market in Egypt: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (USD Million)
- Figure 26.19 Connected Drug Delivery Devices Market in Israel: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (USD Million)
- Figure 26.20 Connected Drug Delivery Devices Market in Saudi Arabia: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (USD Million)
- Figure 26.21 Connected Drug Delivery Devices Market in Rest of Middle East and North Africa: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (USD Million)
- Figure 26.22 Connected Drug Delivery Devices Market in Latin America and Rest of the World: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (Million Units Sold, USD Million)
- Figure 26.23 Connected Drug Delivery Devices Market in Brazil: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (USD Million)
- Figure 26.24 Connected Drug Delivery Devices Market in Argentina: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (USD Million)
- Figure 26.25 Connected Drug Delivery Devices Market in Other Countries: Historical Trends (since 2020) and Forecasted Estimates (till 2035) (USD Million)
- Figure 27.1 Concluding Remarks: Connected Drug Delivery Devices Market Landscape
- Figure 27.2 Concluding Remarks: Add-on-Sensors Market Landscape
- Figure 27.3 Concluding Remarks: Partnerships and Collaborations
- Figure 27.4 Concluding Remarks: Funding and Investments
- Figure 27.5 Concluding Remarks: Market Sizing and Opportunity Analysis











