 |
市場調查報告書
商品編碼
1911394
藥物噴霧乾燥:市場佔有率分析、產業趨勢與統計、成長預測(2026-2031)Pharmaceutical Spray Drying - Market Share Analysis, Industry Trends & Statistics, Growth Forecasts (2026 - 2031) |
||||||
※ 本網頁內容可能與最新版本有所差異。詳細情況請與我們聯繫。
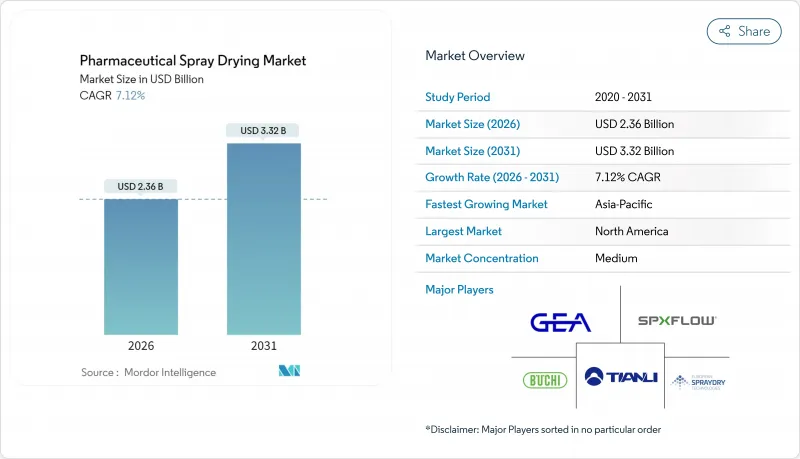
2025 年,醫藥噴霧乾燥市場價值為 22 億美元,預計從 2026 年的 23.6 億美元成長到 2031 年的 33.2 億美元,在預測期(2026-2031 年)內,複合年成長率為 7.12%。
這一發展勢頭使藥物噴霧乾燥市場成為現代藥物研發策略的核心驅動力,這些策略優先考慮提高藥物溶解度、加快生物利用度和實現可擴展的連續流生產。推動成長的因素包括:有利的監管政策、對先進固體分散體日益成長的需求,以及合約研發生產機構 (CDMO) 向整合顆粒工程服務的顯著轉變。設備供應商正在整合即時分析和先進的自動化技術,而生物製藥開發商則利用這些平台來縮短小分子原料藥、疫苗、生物製藥和吸入療法的研發週期。永續性需求、溶劑減量要求以及不斷提高的能源效率目標,進一步加速了閉合迴路和熔融分散系統的應用,鞏固了藥物噴霧乾燥市場作為下一代治療藥物首選生產途徑的地位。
全球製藥噴霧乾燥市場趨勢及洞察
提高藥物溶解度的需求日益成長
約40%的在開發平臺水溶性較差,因此噴霧乾燥無定形固體分散體對於釋放其治療潛力至關重要。臨床試驗表明,該方法可將灰黃黴素的溶出時間從21.5分鐘縮短至8.5分鐘,生物利用度可望提高三倍。共聚維酮和羥丙基甲基纖維素基質可維持胃腸道液中的過飽和狀態,而乙酸處理可使弱鹼的溶解度提高10倍,同時保持相當的體內表現。這些技術的進步持續推動藥物噴霧乾燥市場的強勁需求。
藥品製劑服務外包增加
製藥業對專業CDMO的依賴程度日益加深。 Catalent與賽諾菲活性成分解決方案公司的合作確保了其能夠獲得Niro PSD2/PSD4乾燥設備,從而在無需大量資本投入的情況下實現快速規模化生產。 Hovione的ViSync業務將細胞和基因技術與顆粒工程相結合,標誌著CDMO正式進軍治療方法多元化。隨著連續加工技術的普及,外包降低了驗證風險,並加快了監管申報速度,從而推動了藥物噴霧乾燥市場的成長。
高昂的初始資本投資和營運成本
安裝製藥級噴霧乾燥設備的成本在200萬美元至1000萬美元之間,其中包括高效能空氣過濾器(HEPA過濾器)、隨線分析和10萬級潔淨室。年消費量可達每噸2500至4000千瓦時,從而推高營運預算。此外,維護和GMP認證還會增加20%至25%的經常性間接成本。諸如BUCHI的S-300等提高產量的技術創新可以部分抵消這些成本。然而,中小型贊助商通常選擇與合約研發生產機構(CDMO)合作,在維持製藥噴霧乾燥市場產能的同時,減少直接設備投資。
細分市場分析
預計到2025年,輔料生產將佔總收入的42.85%,這充分體現了噴霧乾燥技術在生產具有流動性、可壓縮性和直接壓片性的粉末方面的優勢,從而提高了片劑生產的效率。兼具崩壞和黏合性的多功能輔料可減少配方步驟,並提高生產柔軟性。生物利用度增強應用預計將以9.05%的複合年成長率成長,這反映了業界對利用具有治療價值但溶解度低的分子的關注。
掩味、緩釋和包封仍然是重要的細分領域。羅蓋特公司於2024年收購IFF Pharma Solutions,凸顯了製劑創新在策略上的重要性,它將聚合物科學與噴霧乾燥技術相結合,以應對不斷變化的給藥挑戰。這些應用領域的趨勢表明,藥物噴霧乾燥市場將持續擴張。
到2025年,小分子原料藥將佔總收入的47.10%。各公司正在採用這項技術將結晶質活性成分轉化為無定形形式,確保快速溶解和體內持續暴露。疫苗,包括mRNA/LNP製劑,將成為成長最快的類別,複合年成長率將達到9.62%。疫情期間的投資驗證了常溫保存的噴霧乾燥吸入疫苗的有效性,使其能夠擺脫冷藏鏈的限制,實現全球分銷。
生物製藥正受益於能夠保留其三級結構的溫和乾燥技術,其中單株抗體的粉末回收率超過90%,且穩定性指標與冷凍乾燥製劑相當。肽類和基因治療的應用案例日益增多,進一步增強了藥物噴霧乾燥市場在各個治療領域的適用性。
區域分析
北美將在2025年繼續保持領先地位,市場佔有率將達到39.85%,這主要得益於其大規模的合約研發生產能力、美國食品藥物管理局(FDA)支持創新的態度以及大型製藥企業的大規模資本投資。 Catalent公司在肯塔基州工廠投資4,000萬美元進行擴建,以及Seran Biosciences公司在佛羅裡達州本德市工廠計劃2億美元,都是持續資本投資的例證。
預計到2031年,亞太地區將以8.18%的最高成長速度成長,主要得益於中國、韓國和印度產能的積極擴張。 SK Pharmatech在韓國投資2.6億美元的工廠體現了與半導體產業同等的投資理念。政府對先進製造技術的支持以及簡化的法規結構正在加速本地化應用,為製藥噴霧乾燥市場的發展提供了強勁動力。
歐洲擁有強大的製造地,在歐盟「地平線計畫」的津貼下,相關系統正在不斷改進。勃林格殷格翰在希臘投資1.2億歐元擴建工廠,將整合新型分子的噴霧乾燥粉末生產。同時,在世界衛生組織技術轉移舉措和各國衛生政策改革的推動下,中東、非洲和南美地區正從依賴進口的地區轉型為區域製劑中心。
其他福利:
- Excel格式的市場預測(ME)表
- 3個月的分析師支持
目錄
第1章 引言
- 研究假設和市場定義
- 調查範圍
第2章調查方法
第3章執行摘要
第4章 市場情勢
- 市場概覽
- 市場促進因素
- 提高藥物溶解度的需求日益成長
- 藥品製劑服務外包增加
- 擴大吸入式和口服薄膜療法
- 連續加工技術的應用日益廣泛
- 對先進藥物輸送平台的有利監管支持
- 市場限制
- 高昂的初始資本投資和營運成本
- 嚴格的驗證和合規要求
- 噴霧乾燥級添加劑供應有限
- 能源強度和永續性問題
- 監管環境
- 波特五力模型
- 新進入者的威脅
- 買方的議價能力
- 供應商的議價能力
- 替代品的威脅
- 競爭對手之間的競爭
第5章 市場規模與成長預測
- 透過使用
- 積層製造
- 封裝
- 提高生物有效性
- 其他用途
- 依配方類型
- 小分子原料藥
- 生物製藥和胜肽類
- 疫苗(包括mRNA/LNP)
- 吸入劑
- 其他
- 噴霧乾燥型
- 旋轉霧化器
- 噴嘴霧化器
- 流體化床/封閉回路型
- 其他
- 按尺寸
- 實驗室/中試規模
- 商業規模
- 地區
- 北美洲
- 美國
- 加拿大
- 墨西哥
- 歐洲
- 德國
- 英國
- 法國
- 義大利
- 西班牙
- 其他歐洲地區
- 亞太地區
- 中國
- 日本
- 印度
- 澳洲
- 韓國
- 亞太其他地區
- 中東和非洲
- GCC
- 南非
- 其他中東和非洲地區
- 南美洲
- 巴西
- 阿根廷
- 南美洲其他地區
- 北美洲
第6章 競爭情勢
- 市場集中度
- 市佔率分析
- 公司簡介
- GEA Group AG
- SPX FLOW Inc.
- Buchi Labortechnik AG
- ProCepT NV
- European SprayDry Technologies
- Shandong Tianli Energy
- Labplant UK
- New AVM Systech
- Advanced Drying Systems
- Lemar Drying Engineering
- Freund-Vector Corp.
- Glatt GmbH
- Fluid Air(Spraying Systems)
- Syntegon Technology
- Spray-Tek Inc.
- Coperion GmbH
- Carrier Vibrating Equipment
- Powder Systems Ltd.
- Innojet GmbH
- Cyclops Spray Drying
第7章 市場機會與未來展望
The pharmaceutical spray drying market was valued at USD 2.20 billion in 2025 and estimated to grow from USD 2.36 billion in 2026 to reach USD 3.32 billion by 2031, at a CAGR of 7.12% during the forecast period (2026-2031).

This momentum positions the pharmaceutical spray drying market size as a core enabler for contemporary drug development strategies that prioritize enhanced solubility, rapid bioavailability, and scalable continuous-flow production. Growth is fueled by supportive regulation, rising demand for advanced solid dispersions, and a clear pivot by contract development and manufacturing organizations (CDMOs) toward integrated particle-engineering services. Equipment suppliers are embedding real-time analytics and advanced automation, while biopharma innovators leverage the platform to shorten development cycles for small-molecule APIs, vaccines, biologics, and inhalable therapies. Sustainability imperatives, solvent-reduction mandates, and heightened energy-efficiency objectives further accelerate adoption of closed-loop and fusion-dispersion systems, reinforcing the pharmaceutical spray drying market as a preferred route for next-generation therapeutics.
Global Pharmaceutical Spray Drying Market Trends and Insights
Growing Demand for Enhanced Drug Solubility
Approximately 40 % of pipeline molecules present low aqueous solubility, making spray-dried amorphous solid dispersions indispensable for recovery of therapeutic potential. Clinically, the method reduced griseofulvin dissolution times from 21.5 minutes to 8.5 minutes, tripling bioavailability improvement prospects. Copovidone and hypromellose matrices sustain supersaturation in gastrointestinal fluids, and acetic-acid aided processing elevates solubility of weak bases tenfold while maintaining equivalent in-vivo performance. Together, these advances sustain robust forward demand for the pharmaceutical spray drying market.
Rising Outsourcing of Pharmaceutical Formulation Services
Pharma's reliance on specialty CDMOs is intensifying. Catalent's collaboration with Sanofi Active Ingredient Solutions secured access to Niro PSD2/PSD4 dryers, enabling rapid scale without significant capital outlay. Hovione's ViSync venture pairs cell-and-gene technologies with particle engineering, illustrating CDMOs' push into modality diversification. As continuous processing skills proliferate, outsourcing mitigates validation risk and accelerates dossier filings, anchoring growth in the pharmaceutical spray drying market.
High Initial Capital Investment and Operating Costs
Pharmaceutical-grade spray dryer installations range from USD 2-10 million inclusive of HEPA filtration, in-line analytics, and Class 100,000 cleanrooms. Annual energy consumption of 2,500-4,000 kWh per ton inflates operating budgets, while maintenance and GMP certification add 20-25 % repeating overhead. Innovations such as BUCHI's S-300, with enhanced yield capture, partially offset these burdens. Nonetheless, smaller sponsors often pivot to CDMOs, reducing direct equipment uptake yet sustaining volumes within the pharmaceutical spray drying market.
Other drivers and restraints analyzed in the detailed report include:
- Expansion of Inhalable and Oral Thin-Film Therapies
- Increasing Adoption of Continuous Processing Technologies
- Stringent Validation and Compliance Requirements
For complete list of drivers and restraints, kindly check the Table Of Contents.
Segment Analysis
Excipient production held 42.85 % of 2025 revenue, highlighting spray drying's ability to engineer flowable, compressible, and directly compressible powders that streamline tablet manufacturing. Spray-dried multifunctional excipients with integrated disintegration and binding properties reduce formulation steps, enhancing manufacturing agility. Bioavailability-enhancement applications are forecast to advance at a 9.05 % CAGR, reflecting industry emphasis on unlocking therapeutically valuable but poorly soluble molecules.
Taste-masking, controlled-release, and encapsulation remain vital niches. Roquette's acquisition of IFF Pharma Solutions in 2024 underscores the strategic importance of excipient innovation, combining polymer science with spray-drying know-how to address evolving delivery challenges. Collectively, the application landscape signals sustained expansion of the pharmaceutical spray drying market.
Small-molecule APIs provided 47.10 % of 2025 revenue. Companies deploy the technology to transform crystalline actives into amorphous forms, securing rapid dissolution and consistent in-vivo exposure. Vaccines, including mRNA/LNP formats, represent the fastest-growing category with a projected 9.62 % CAGR. Pandemic-era investments validated room-temperature, spray-dried respirable vaccines that facilitate global distribution without cold chain constraints.
Biologics benefit from gentle drying that preserves tertiary structure, with monoclonal antibodies achieving > 90 % powder recovery and stability metrics comparable to lyophilized alternatives. Peptide and gene-therapy use cases are gaining traction, reinforcing the adaptability of the pharmaceutical spray drying market across therapeutic classes.
The Pharmaceutical Spray Drying Market Report is Segmented by Application (Excipient Production, and More), Formulation Type (Small-Molecule APIs, and More), Spray Dryer Type (Rotary Atomizer, and More), Scale (Laboratory/Pilot and Commercial-Scale), and Geography (North America, Europe, Asia-Pacific, Middle East & Africa, South America). The Market Forecasts are Provided in Terms of Value (USD).
Geography Analysis
North America maintained leadership at 39.85 % share in 2025, cemented by substantial CDMO capacity, a pro-innovation FDA stance, and large-scale internal builds from big pharma. Catalent's USD 40 million Kentucky upgrade and Seran BioScience's USD 200 million Bend project illustrate sustained capital deployment.
Asia-Pacific will post the fastest 8.18 % CAGR through 2031, driven by aggressive capacity expansion across China, South Korea, and India. SK pharmteco's USD 260 million South Korean facility exemplifies the region's semiconductor-like investment mentality. Government incentives for advanced manufacturing and simplified regulatory frameworks accelerate local uptake, injecting tailwinds into the pharmaceutical spray drying market.
Europe remains a stalwart manufacturing cluster, adding continuous systems under Horizon Europe grants. Boehringer Ingelheim's EUR 120 million Greek expansion integrates spray-dried powder production for novel molecules. Meanwhile, Middle East & Africa and South America are evolving from import-dependent regions into regional formulation hubs, encouraged by WHO tech-transfer initiatives and domestic healthcare policy reforms.
- GEA Group
- SPX FLOW Inc.
- Buchi Labortechnik AG
- ProCepT NV
- European SprayDry Technologies
- Shandong Tianli Energy
- Labplant UK
- New AVM Systech
- Advanced Drying Systems
- Lemar Drying Engineering
- Freund-Vector Corp.
- Glatt
- Fluid Air (Spraying Systems)
- Syntegon Technology
- Spray-Tek Inc.
- Coperion
- Carrier Vibrating Equipment
- Powder Systems Ltd.
- Innojet GmbH
- Cyclops Spray Drying
Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
TABLE OF CONTENTS
1 Introduction
- 1.1 Study Assumptions & Market Definition
- 1.2 Scope of the Study
2 Research Methodology
3 Executive Summary
4 Market Landscape
- 4.1 Market Overview
- 4.2 Market Drivers
- 4.2.1 Growing Demand for Enhanced Drug Solubility
- 4.2.2 Rising Outsourcing of Pharmaceutical Formulation Services
- 4.2.3 Expansion of Inhalable and Oral Thin-Film Therapies
- 4.2.4 Increasing Adoption of Continuous Processing Technologies
- 4.2.5 Favorable Regulatory Support for Advanced Drug Delivery Platforms
- 4.3 Market Restraints
- 4.3.1 High Initial Capital Investment and Operating Costs
- 4.3.2 Stringent Validation and Compliance Requirements
- 4.3.3 Limited Availability of Spray-Drying-Grade Excipients
- 4.3.4 Energy Intensity and Sustainability Concerns
- 4.4 Regulatory Landscape
- 4.5 Porter's Five Forces
- 4.5.1 Threat of New Entrants
- 4.5.2 Bargaining Power of Buyers
- 4.5.3 Bargaining Power of Suppliers
- 4.5.4 Threat of Substitutes
- 4.5.5 Competitive Rivalry
5 Market Size & Growth Forecasts (Value, USD)
- 5.1 By Application
- 5.1.1 Excipient Production
- 5.1.2 Encapsulation
- 5.1.3 Enhancing Bioavailability
- 5.1.4 Other Applications
- 5.2 By Formulation Type
- 5.2.1 Small-Molecule APIs
- 5.2.2 Biologics & Peptides
- 5.2.3 Vaccines (Incl. mRNA/LNP)
- 5.2.4 Inhalable Formulations
- 5.2.5 Others
- 5.3 By Spray Dryer Type
- 5.3.1 Rotary Atomizer
- 5.3.2 Nozzle Atomizer
- 5.3.3 Fluidized / Closed-Loop
- 5.3.4 Others
- 5.4 By Scale
- 5.4.1 Laboratory / Pilot
- 5.4.2 Commercial-Scale
- 5.5 Geography
- 5.5.1 North America
- 5.5.1.1 United States
- 5.5.1.2 Canada
- 5.5.1.3 Mexico
- 5.5.2 Europe
- 5.5.2.1 Germany
- 5.5.2.2 United Kingdom
- 5.5.2.3 France
- 5.5.2.4 Italy
- 5.5.2.5 Spain
- 5.5.2.6 Rest of Europe
- 5.5.3 Asia-Pacific
- 5.5.3.1 China
- 5.5.3.2 Japan
- 5.5.3.3 India
- 5.5.3.4 Australia
- 5.5.3.5 South Korea
- 5.5.3.6 Rest of Asia-Pacific
- 5.5.4 Middle East & Africa
- 5.5.4.1 GCC
- 5.5.4.2 South Africa
- 5.5.4.3 Rest of Middle East & Africa
- 5.5.5 South America
- 5.5.5.1 Brazil
- 5.5.5.2 Argentina
- 5.5.5.3 Rest of South America
- 5.5.1 North America
6 Competitive Landscape
- 6.1 Market Concentration
- 6.2 Market Share Analysis
- 6.3 Company Profiles (includes Global level Overview, Market level overview, Core Business Segments, Financials, Headcount, Key Information, Market Rank, Market Share, Products and Services, and analysis of Recent Developments)
- 6.3.1 GEA Group AG
- 6.3.2 SPX FLOW Inc.
- 6.3.3 Buchi Labortechnik AG
- 6.3.4 ProCepT NV
- 6.3.5 European SprayDry Technologies
- 6.3.6 Shandong Tianli Energy
- 6.3.7 Labplant UK
- 6.3.8 New AVM Systech
- 6.3.9 Advanced Drying Systems
- 6.3.10 Lemar Drying Engineering
- 6.3.11 Freund-Vector Corp.
- 6.3.12 Glatt GmbH
- 6.3.13 Fluid Air (Spraying Systems)
- 6.3.14 Syntegon Technology
- 6.3.15 Spray-Tek Inc.
- 6.3.16 Coperion GmbH
- 6.3.17 Carrier Vibrating Equipment
- 6.3.18 Powder Systems Ltd.
- 6.3.19 Innojet GmbH
- 6.3.20 Cyclops Spray Drying
7 Market Opportunities & Future Outlook
- 7.1 White-Space & Unmet-Need Assessment






