 |
市場調查報告書
商品編碼
1961003
全球CAR-T細胞療法市場:按產品、標靶、適應症、人口統計特徵、最終用戶和地區分類-預測至2031年CAR T-Cell Therapy Market by Product (Abecma, Breyanzi, Carvykti, Yescarta, Tecartus), Target (CD19, BCMA), Indication (Multiple Myeloma, Leukemia, Lymphoma), Demographic (Adult, Pediatric), Region, Competitive Landscape - Global Forecast to 2031 |
||||||
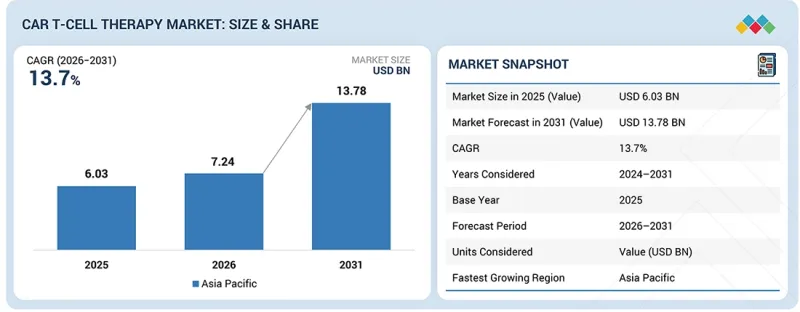
預計 CAR T 細胞療法的全球市場規模將從 2026 年的 72.4 億美元成長到 2031 年的 137.8 億美元,預計複合年成長率為 13.7%。
全球癌症發生率的上升是市場成長的主要促進因素。 CAR-T細胞療法的技術進步以及治療方法研發領域投資和資金籌措的增加,預計將進一步推動市場成長。
| 調查範圍 | |
|---|---|
| 調查期 | 2024-2031 |
| 基準年 | 2025 |
| 預測期 | 2026-2031 |
| 目標單元 | 金額(10億美元) |
| 部分 | 產品、目標族群、適應症、人口統計資料、最終使用者、地區 |
| 目標區域 | 北美、歐洲、亞太地區、拉丁美洲、中東和非洲 |
然而,CAR T 細胞療法的副作用和高昂的治療費用預計將成為預測期內限制市場成長的主要因素。
依產品分類,CAR T 細胞療法市場主要分為以下幾類產品:Abekuma(idekabutagene/vicreucel)、Breyanzi(lysokabutagene/malareucel)、Calvicti(siltakabutagene/autoleucel)、Yeskata(axikabutagene/silaucel)、Teamah 和其他產品(Acelnone)。
2025年,Jeskarta佔據了最大的市場。市場成長得益於Jeskarta在治療復發/難治性癌症方面的高緩解率和持續緩解效果,從而推動了治療方法的普及。
從終端使用者角度來看,全球CAR-T細胞療法市場可細分為醫院、長期照護機構和專科治療中心。到2025年,醫院領域將成為全球CAR-T細胞療法市場的主要驅動力,並在預測期內保持最高的複合年成長率。
醫院正將CAR-T細胞療法納入其標準腫瘤治療通訊協定。隨著越來越多的醫院採用CAR-T細胞療法,這種趨勢正在推動市場需求。此外,醫院在加速CAR-T細胞療法的臨床試驗中發揮著至關重要的作用。積極參與研究舉措以及與製藥公司和醫院的合作,能夠擴大依證,加速監管核准,從而進一步推動市場成長。
CAR-T細胞療法市場按地區分類為北美、歐洲、亞太、拉丁美洲以及中東和非洲。預計亞太地區在預測期內將以最高的複合年成長率成長。有利的法規結構、豐富的CAR-T細胞療法產品平臺以及加速核准流程將促進創新治療方法進入市場,並推動亞太地區的成長。此外,中國醫院、大學和產業界之間日益緊密的合作預計也將推動市場強勁成長。在中國,CAR-T細胞療法在多種癌症類型中的安全性和有效性評估臨床試驗數量迅速增加,也取得了顯著進展。這些研究在治療白血病、淋巴瘤和固態腫瘤方面顯示出令人鼓舞的結果,在特定患者群體中,完全緩解率和持續緩解率顯著提高。這一趨勢凸顯了亞太地區在CAR-T細胞療法的研發和檢驗方面的進展,並將在改善癌症治療效果方面發揮關鍵作用。
本報告涵蓋的公司簡介清單:
- 百時美施貴寶公司(美國)
- 吉利德科學公司(美國)
- 諾華公司(瑞士)
- 強生公司(美國)
- 卡瑞生製藥控股股份有限公司(中國)
- IASO生物治療(中國)
- JW(開曼)治療(中國)
- ImmunoAct (India)
- CRISPR Therapeutics(瑞士)
- Autolus Therapeutics(英國)
- Allogene Therapeutics(美國)
- Cartesian Therapeutics Inc.(美國)
- 廣州百奧基因科技有限公司(中國)
- Wugen(美國)
購買本報告的主要好處:
本報告透過提供CAR-T細胞療法市場及其細分領域最精準的收入預測,協助市場領導和新參與企業。報告也幫助相關人員深入了解競爭格局,更有效地進行市場定位,並制定合適的打入市場策略。此外,本報告還能幫助相關人員掌握市場趨勢,並取得關鍵市場促進因素、限制因素、機會和挑戰的資訊。
本報告深入分析了以下幾點:
對關鍵促進因素(CAR-T 細胞療法的技術進步、由於血液惡性腫瘤的強療效訊號而擴大的適應症以及對治療方法研發投入的增加)、阻礙因素(高成本和報銷挑戰)、機遇(從骨髓惡性腫瘤擴展到固態腫瘤和新興疾病領域,以及將重點轉向同種異體 CAR-T 和 CAR-NKNK 的變化分析的變化性 (CAR-NK 和 TCAR 分析
- 產品開發與創新:對 CAR T 細胞療法市場的新產品發布進行詳細分析。
- 市場發展:盈利市場的全面資訊-本報告分析了各個地區的 CAR T 細胞療法市場。
- 市場多元化:全面介紹 CAR T 細胞療法市場的新產品、未開發的市場、近期趨勢和投資情況。
目錄
第1章:引言
第2章執行摘要
第3章重要考察
第4章 市場概覽
- 市場動態
- 未滿足的需求和閒置頻段
- 相互關聯的市場與跨產業機遇
- 1/2/3級玩家的策略舉措
- 管道分析
第5章 產業趨勢
- 波特五力分析
- 宏觀經濟展望
- 價值鏈分析
- 生態系分析
- 價格分析
- 2026-2027 年主要會議和活動
- 影響客戶業務的趨勢/顛覆性因素
- 投資/資金籌措方案
- 2025年美國關稅對自動化CAR-T細胞療法市場的影響
第6章:技術進步、人工智慧的影響、專利、創新與未來應用
- 技術分析
- 技術/產品藍圖
- 未來應用
- 人工智慧/生成式人工智慧對CAR-T細胞療法市場的影響
第7章永續性和監管情勢
- 當地法規和合規性
- 對永續性和監管政策措施的影響
- 認證、標籤和環境標準
- 救贖方案
- 聯合治療
第8章:顧客趨勢與購買行為
- 決策流程
- 買方相關人員和採購評估標準
- 招募障礙和內部挑戰
- 來自各個終端使用者產業的未滿足需求
- 市場盈利
第9章 CAR-T細胞療法市場(依產品分類)
- 阿維克瑪
- 布雷揚格
- 卡爾維克蒂
- 耶穌·卡塔
- 泰卡圖斯
- 金·裡亞
- 其他
第10章 CAR-T細胞療法市場(依目標市場分類)
- CD19
- BCMA
- CD19/20
- CD7
- 其他
第11章:CAR-T細胞療法市場(依適應症分類)
- B細胞淋巴瘤(BCL)
- 多發性骨髓瘤
- 急性淋巴性白血病(全部)
- 其他
第12章 CAR-T細胞療法市場(依人口統計資料分類)
- 成人
- 兒童
第13章:CAR-T細胞療法市場(以最終用戶分類)
- 醫院
- 專業中心
- 長期照護機構
第14章:CAR-T細胞療法市場(按地區分類)
- 北美洲
- 美國
- 加拿大
- 歐洲
- 德國
- 英國
- 法國
- 義大利
- 西班牙
- 其他
- 亞太地區
- 中國
- 日本
- 印度
- 澳洲
- 韓國
- 其他
- 拉丁美洲
- 巴西
- 其他
- 中東
- 海灣合作理事會國家
- 沙烏地阿拉伯王國
- 阿拉伯聯合大公國
- 其他
- 非洲
- 血液系統惡性腫瘤和感染疾病帶來的沉重負擔正在推動市場成長。
第15章 競爭格局
- 主要參與企業的策略/優勢
- 2023-2025年收入分析
- 2025年市佔率分析
- 企業估值矩陣:主要參與企業,2025 年
- 公司估值矩陣:Start-Ups/中小企業,2025 年
- 企業估值和財務指標
- 品牌/產品對比
- 競爭格局
第16章:公司簡介
- 主要參與企業
- JOHNSON & JOHNSON
- GILEAD SCIENCES, INC.
- BRISTOL-MYERS SQUIBB COMPANY
- NOVARTIS AG
- AUTOLUS THERAPEUTICS
- JW(CAYMAN)THERAPEUTICS CO., LTD.
- IMMUNOADOPTIVE CELL THERAPY PRIVATE LIMITED(IMMUNOACT)
- CARSGEN THERAPEUTICS HOLDINGS LIMITED
- IASO BIOTHERAPEUTICS
- IMMUNEEL THERAPEUTICS
- 其他公司
- WUGEN
- CARTESIAN THERAPEUTICS, INC.
- ALLOGENE THERAPEUTICS
- LYELL IMMUNOPHARMA, INC.
- KYVERNA THERAPEUTICS, INC.
- CELLICTIS SA
- BRAINCHILD BIO
- ATARA BIOTHERAPEUTICS, INC.
- CARIBOU BIOSCIENCES
- ARCELLX
- CRISPR THERAPEUTICS
- POSEIDA THERAPEUTICS
- CABALETTA BIO
第17章調查方法
第18章附錄
The global CAR T-cell therapy market is projected to reach USD 13.78 billion in 2031 from USD 7.24 billion in 2026, at a CAGR of 13.7%. Market growth is primarily driven by rising global cancer prevalence. Technological advancements in CAR T-cell therapies and rising investment and funding for therapy development are likely to propel market growth.
| Scope of the Report | |
|---|---|
| Years Considered for the Study | 2024-2031 |
| Base Year | 2025 |
| Forecast Period | 2026-2031 |
| Units Considered | Value (USD billion) |
| Segments | By Product | By Target | By Indication | By Demographic | By End user |
| Regions covered | North America, Europe, Asia Pacific, Latin America, Middle East and Africa |
However, adverse effects associated with CAR T-cell therapy and high treatment costs are among the major factors expected to restrain market growth during the forecast period.

"The Yescarta product segment accounted for the largest share by product in the CAR T-cell therapy market in 2025."
Based on product, the CAR T-cell therapy market is broadly segmented into Abecma (idecabtagene vicleucel), Breyanzi (lisocabtagene maraleucel), Carvykti (ciltacabtagene autoleucel), Yescarta (axicabtagene ciloleucel), Tecartus (brexucabtagene autoleucel), Kymriah (tisagenlecleucel), and other products.
The Yescarta segment held the largest market share in 2025. Market growth is driven by high response rates and durable remissions with Yescarta in treating relapsed/refractory cancers, leading to increased therapy adoption.
"The hospitals segment accounted for the largest share by end user segment in the CAR T-cell therapy market in 2025."
Based on end users, the global CAR T-cell therapy market is segmented into hospitals, long-term care facilities, and specialty centers. In 2025, the hospitals segment emerged as the primary growth driver in the global CAR T-cell therapy market, with the highest CAGR during the forecast period.
Hospitals are incorporating CAR T-cell therapies into their standard oncology treatment protocols. This trend is boosting demand for CAR T-cell therapies as more hospitals embrace them. Furthermore, hospitals play a key role in facilitating clinical trials for CAR T-cell therapies. By actively participating in research initiatives and collaborating with pharmaceutical companies and hospitals, the evidence base expands, and regulatory approvals accelerate, which, in turn, stimulate market growth.
"Asia Pacific is projected to grow at the highest CAGR in the CAR T-cell therapy market from 2026 to 2031."
The CAR T-cell therapy market is segmented into North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa. During the forecast period, Asia Pacific is estimated to grow at the highest CAGR. The supportive regulatory frameworks, rich CAR T-cell therapy product pipeline, and expedited approval processes facilitate quicker market entry for innovative therapies, driving growth in Asia Pacific. In addition, increased collaborations among hospitals, universities, and industries in China are expected to drive robust market growth. The rapid increase in clinical trials evaluating the safety and effectiveness of CAR T-cell therapy across diverse cancer types in China also signals significant advancements. These studies have demonstrated encouraging results in treating leukemia, lymphoma, and solid tumors, with specific patient cohorts showing substantial rates of complete remission and sustained responses. This trend highlights Asia Pacific's progress in developing and validating CAR T-cell therapies, positioning the region as a key player in advancing oncology therapeutic outcomes.
The primary interviews conducted for this report can be categorized as follows:
- By Company Type: Tier 1- 40%, Tier 2- 30%, and Tier 3- 30%
- By Designation: Directors- 20%, Managers- 10%, and Others - 70%,
- By Region: North America -35%, Europe -25%, Asia Pacific -25%, Latin America -10%, and Middle East -5%.
List of Companies Profiled in the Report:
- Bristol-Myers Squibb Company (US)
- Gilead Sciences Inc. (US)
- Novartis AG (Switzerland)
- Johnson & Johnson (US)
- CARsgen Therapeutics Holdings Limited (China)
- IASO Biotherapeutics (China)
- JW (Cayman) Therapeutics Co., Ltd (China)
- ImmunoAct (India)
- CRISPR Therapeutics (Switzerland)
- Autolus Therapeutics (UK)
- Allogene Therapeutics (US)
- Cartesian Therapeutics Inc. (US)
- Guangzhou Bio-gene Technology Co., Ltd (China)
- Wugen (US)
Research Coverage:
This research report categorizes the CAR T-cell therapy market by product (YESCARTA, KYMRIAH, CARVYKTI, ABECMA, TECARTUS, BREYANZI, Other products); by target: CD19, BCMA, other targets; by indication: Multiple myeloma, B-cell lymphoma, Acute lymphoblastic leukemia, Other indications; by demographics: adult, pediatric; by End User: Hospitals, Specialty centers, Long-term care facilities, and by region: North America, Europe, Asia Pacific, Latin America, Middle East & Africa.
The scope of the report covers detailed information regarding the major factors, such as drivers, challenges, opportunities, and restraints, influencing the growth of the CAR T-cell therapy market. A detailed analysis of key industry players has been conducted to provide insights into their business overview, product portfolios, key strategies, such as product launches, collaborations, partnerships, expansions, agreements, and recent developments in the CAR T-cell therapy market. This report provides a competitive analysis of top players and emerging startups in the CAR T-cell therapy market ecosystem.
Key Benefits of Buying the Report:
The report will help market leaders/new entrants by providing the closest approximations of revenue for the overall CAR T-cell therapy market and its subsegments. It will also help stakeholders better understand the competitive landscape and gain more insights to position their business more effectively and develop suitable go-to-market strategies. This report will enable stakeholders to understand the market's pulse and provide them with information on the key market drivers, restraints, opportunities, and challenges.
The report provides insights into the following pointers:
Analysis of key drivers (Technological advancements in CAR T-cell therapies, Label expansions along with strong efficacy signals in hematologic cancers, Growing investments in the development of therapies), restraints (High cost and reimbursement challenges), opportunities (Expanding beyond hematologic malignancies into solid tumors and new disease areas, Focus shift on allogeneic CAR-T and CAR-NK), and challenges (Long-term safety surveillance and evolving labeling requirements) are influencing the growth of CAR T-cell therapy market.
- Product Development/Innovation: Detailed insights on newly launched products of the CAR T-cell therapy market.
- Market Development: Comprehensive information about lucrative markets - the report analyses the CAR T-cell therapy market across varied regions.
- Market Diversification: Exhaustive information about new products, untapped geographies, recent developments, and investments in the CAR T-cell therapy market.
- Competitive Assessment: In-depth assessment of market shares, growth strategies, and product offerings of leading players include Bristol-Myers Squibb Company (US), Gilead Sciences, Inc. (US), Novartis AG (Switzerland), Johnson & Johnson (US), JW (Cayman) Therapeutics Co. Ltd (China), ImmunoAct (India), among others in the CAR T-cell therapy market.
TABLE OF CONTENTS
1 INTRODUCTION
- 1.1 STUDY OBJECTIVES
- 1.2 MARKET DEFINITION
- 1.3 STUDY SCOPE
- 1.3.1 MARKET SEGMENTATION & REGIONAL SCOPE
- 1.3.2 INCLUSIONS & EXCLUSIONS
- 1.3.3 YEARS CONSIDERED
- 1.3.4 CURRENCY CONSIDERED
- 1.4 STAKEHOLDERS
- 1.5 SUMMARY OF CHANGES
2 EXECUTIVE SUMMARY
- 2.1 KEY INSIGHTS & MARKET HIGHLIGHTS
- 2.2 KEY MARKET PARTICIPANTS: SHARE INSIGHTS AND STRATEGIC DEVELOPMENTS
- 2.3 DISRUPTIVE TRENDS SHAPING MARKET
- 2.4 HIGH-GROWTH SEGMENTS & EMERGING FRONTIERS
- 2.5 SNAPSHOT: GLOBAL MARKET SIZE, GROWTH RATE, AND FORECAST
3 PREMIUM INSIGHTS
- 3.1 CAR T-CELL THERAPY MARKET OVERVIEW
- 3.2 NORTH AMERICA: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC AND COUNTRY, 2025
- 3.3 CAR T-CELL THERAPY MARKET: GEOGRAPHIC SNAPSHOT
4 MARKET OVERVIEW
- 4.1 INTRODUCTION
- 4.2 MARKET DYNAMICS
- 4.2.1 DRIVERS
- 4.2.1.1 Technological advancements in CAR T-cell therapies
- 4.2.1.2 Label expansions and strong efficacy signals in hematologic cancers
- 4.2.1.3 Growing investment in CAR T-cell therapy development
- 4.2.2 RESTRAINTS
- 4.2.2.1 High cost and reimbursement challenges for CAR T-cell therapy
- 4.2.3 OPPORTUNITIES
- 4.2.3.1 Expanding beyond hematologic malignancies into solid tumors and new disease areas
- 4.2.3.2 Focus shift on allogeneic CAR-T and CAR-NK
- 4.2.3.3 Growing collaborations among biopharmaceutical companies, academic institutions, and research organizations
- 4.2.4 CHALLENGES
- 4.2.4.1 Long-term safety surveillance and evolving labeling requirements
- 4.2.1 DRIVERS
- 4.3 UNMET NEEDS & WHITE SPACES
- 4.4 INTERCONNECTED MARKETS & CROSS-SECTOR OPPORTUNITIES
- 4.5 STRATEGIC MOVES BY TIER-1/2/3 PLAYERS
- 4.6 PIPELINE ANALYSIS
5 INDUSTRY TRENDS
- 5.1 PORTER'S FIVE FORCES ANALYSIS
- 5.1.1 THREAT OF NEW ENTRANTS
- 5.1.2 THREAT OF SUBSTITUTES
- 5.1.3 BARGAINING POWER OF BUYERS
- 5.1.4 BARGAINING POWER OF SUPPLIERS
- 5.1.5 INTENSITY OF COMPETITIVE RIVALRY
- 5.2 MACROECONOMIC OUTLOOK
- 5.2.1 INTRODUCTION
- 5.2.2 GDP TRENDS AND FORECASTS
- 5.2.3 TRENDS IN GLOBAL CAR-T-CELL THERAPY MARKET
- 5.3 VALUE CHAIN ANALYSIS
- 5.4 ECOSYSTEM ANALYSIS
- 5.4.1 ROLE IN ECOSYSTEM
- 5.5 PRICING ANALYSIS
- 5.5.1 AVERAGE SELLING PRICE OF CAR T-CELL THERAPY PRODUCTS, BY TYPE, 2025
- 5.5.2 AVERAGE SELLING PRICE OF CAR T-CELL THERAPY PRODUCTS, BY KEY PLAYER, 2025
- 5.5.3 AVERAGE SELLING PRICE OF CAR T-CELL THERAPY PRODUCTS, BY REGION, 2025
- 5.6 KEY CONFERENCES & EVENTS, 2026-2027
- 5.7 TRENDS/DISRUPTIONS IMPACTING CUSTOMER'S BUSINESS
- 5.8 INVESTMENT/FUNDING SCENARIO
- 5.9 IMPACT OF 2025 US TARIFF ON CAR T-CELL THERAPY MARKET
- 5.9.1 KEY TARIFF RATES
- 5.9.2 PRICE IMPACT ANALYSIS
- 5.9.3 IMPACT ON COUNTRY/REGION
- 5.9.3.1 North America
- 5.9.3.1.1 US
- 5.9.3.2 Europe
- 5.9.3.3 Asia Pacific
- 5.9.3.1 North America
- 5.9.4 IMPACT ON END-USE INDUSTRIES
- 5.9.4.1 Hospitals
- 5.9.4.2 Specialty oncology centers
- 5.9.4.3 Long-term care facilities
6 TECHNOLOGICAL ADVANCEMENTS, AI-DRIVEN IMPACT, PATENTS, INNOVATIONS, AND FUTURE APPLICATIONS
- 6.1 TECHNOLOGY ANALYSIS
- 6.1.1 KEY TECHNOLOGIES
- 6.1.1.1 Car design and optimization
- 6.1.1.2 Viral vector technology
- 6.1.1.3 Cell culture and expansion techniques
- 6.1.2 COMPLEMENTARY TECHNOLOGIES
- 6.1.2.1 Gene editing technology (CRISPR-CAS9-based genome editing)
- 6.1.3 ADJACENT TECHNOLOGIES
- 6.1.3.1 Monitoring and imaging technologies
- 6.1.1 KEY TECHNOLOGIES
- 6.2 TECHNOLOGY/PRODUCT ROADMAP
- 6.3 FUTURE APPLICATIONS
- 6.4 IMPACT OF AI/GEN AI ON CAR T-CELL THERAPY MARKET
7 SUSTAINABILITY AND REGULATORY LANDSCAPE
- 7.1 REGIONAL REGULATIONS AND COMPLIANCE
- 7.1.1 REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- 7.1.2 REGULATORY FRAMEWORK
- 7.1.3 INDUSTRY STANDARDS
- 7.2 SUSTAINABILITY IMPACT AND REGULATORY POLICY INITIATIVES
- 7.3 CERTIFICATIONS, LABELING, AND ECO-STANDARDS
- 7.4 REIMBURSEMENT SCENARIO
- 7.5 COMBINATION THERAPIES
8 CUSTOMER LANDSCAPE & BUYER BEHAVIOR
- 8.1 DECISION-MAKING PROCESS
- 8.2 BUYER STAKEHOLDERS AND BUYING EVALUATION CRITERIA
- 8.2.1 KEY STAKEHOLDERS IN BUYING PROCESS
- 8.2.2 KEY BUYING CRITERIA, BY END USER
- 8.3 ADOPTION BARRIERS & INTERNAL CHALLENGES
- 8.4 UNMET NEEDS FROM VARIOUS END-USE INDUSTRIES
- 8.5 MARKET PROFITABILITY
9 CAR T-CELL THERAPY MARKET, BY PRODUCT
- 9.1 INTRODUCTION
- 9.2 ABECMA
- 9.2.1 FOCUS ON INCREASED RESEARCH ACTIVITIES FOR INDICATION EXPANSIONS TO DRIVE MARKET
- 9.3 BREYANZI
- 9.3.1 NEED FOR TREATMENT OF MULTIPLE INDICATIONS TO PROPEL MARKET GROWTH
- 9.4 CARVYKTI
- 9.4.1 GLOBAL REACH AND SIGNIFICANT REVENUE GROWTH TO SUPPORT MARKET
- 9.5 YESCARTA
- 9.5.1 FOCUS ON REGULATORY APPROVALS ACROSS MAJOR REGIONS TO BOOST MARKET GROWTH
- 9.6 TECARTUS
- 9.6.1 EFFORTS TO SECURE REIMBURSEMENT AND IMPROVE MARKET ACCESS FACILITATE PATIENT TO AID MARKET ADOPTION
- 9.7 KYMRIAH
- 9.7.1 APPROVAL FOR PEDIATRIC USE TO AUGMENT MARKET GROWTH
- 9.8 OTHER PRODUCTS
10 CAR T-CELL THERAPY MARKET, BY TARGET
- 10.1 INTRODUCTION
- 10.2 CD19
- 10.2.1 RISING INDICATION EXPANSIONS AND NEW APPROVALS TO DRIVE MARKET
- 10.3 BCMA
- 10.3.1 GROWING PIPELINE PRODUCTS TO PROPEL MARKET GROWTH
- 10.4 CD19/20
- 10.4.1 DUAL TARGETING TO OFFER COMPETITIVE ADVANTAGE IN ANTIGEN ESCAPE AND TUMOR HETEROGENEITY ISSUES
- 10.5 CD7
- 10.5.1 ALLOGENEIC CELL SOURCE OF CD7 THERAPIES IN DEVELOPMENT TO SUPPORT GROWTH
- 10.6 OTHER TARGETS
11 CAR T-CELL THERAPY MARKET, BY INDICATION
- 11.1 INTRODUCTION
- 11.2 B-CELL LYMPHOMA (BCL)
- 11.2.1 RISING PREVALENCE OF B-CELL LYMPHOMAS TO PROPEL MARKET GROWTH
- 11.3 MULTIPLE MYELOMA
- 11.3.1 GROWING PRODUCT REVENUE AND RISING GLOBAL INCIDENCE OF MYELOMA TO DRIVE MARKET
- 11.4 ACUTE LYMPHOBLASTIC LEUKEMIA (ALL)
- 11.4.1 EXPANDING R&D PIPELINE TO AUGMENT MARKET GROWTH
- 11.5 OTHER INDICATIONS
12 CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC
- 12.1 INTRODUCTION
- 12.2 ADULTS
- 12.2.1 GROWING PREVALENCE OF HEMATOLOGIC CANCERS TO DRIVE MARKET
- 12.3 PEDIATRIC
- 12.3.1 HIGH UNMET NEEDS TO LIMIT SEGMENT GROWTH
13 CAR T-CELL THERAPY MARKET, BY END USER
- 13.1 INTRODUCTION
- 13.2 HOSPITALS
- 13.2.1 GROWING NEED FOR STANDARD ONCOLOGY TREATMENT PROTOCOLS TO DRIVE MARKET
- 13.3 SPECIALTY CENTRES
- 13.3.1 NEED FOR OPTIMAL PATIENT CARE AND SPECIALIZED TREATMENT OPTIONS TO SPUR MARKET GROWTH
- 13.4 LONG-TERM CARE FACILITIES
- 13.4.1 INCREASING INCIDENCE OF CHRONIC DISORDERS AMONG GERIATRIC POPULATION TO PROPEL MARKET GROWTH
14 CAR T-CELL THERAPY MARKET, BY REGION
- 14.1 INTRODUCTION
- 14.2 NORTH AMERICA
- 14.2.1 US
- 14.2.1.1 US to dominate CAR T-cell therapy market during study period
- 14.2.2 CANADA
- 14.2.2.1 Regulatory approvals and government initiatives for regenerative medicine research to drive market
- 14.2.1 US
- 14.3 EUROPE
- 14.3.1 GERMANY
- 14.3.1.1 Key product approvals and focus on clinical research to aid market growth
- 14.3.2 UK
- 14.3.2.1 Technological advancements in automation and presence of robust research infrastructure to spur market growth
- 14.3.3 FRANCE
- 14.3.3.1 Growing focus on cell & gene therapy initiatives to boost market growth
- 14.3.4 ITALY
- 14.3.4.1 Growth in biotech sector to augment market growth
- 14.3.5 SPAIN
- 14.3.5.1 Rising focus on cell therapies and growing focus on stringent regulations to augment market growth
- 14.3.6 REST OF EUROPE
- 14.3.1 GERMANY
- 14.4 ASIA PACIFIC
- 14.4.1 CHINA
- 14.4.1.1 Rising number of CAR T-cell clinical trials to fuel market growth
- 14.4.2 JAPAN
- 14.4.2.1 Increasing number of product approvals and rising geriatric population to propel market growth
- 14.4.3 INDIA
- 14.4.3.1 Growing focus on product commercialization and rising cancer incidence to boost market demand
- 14.4.4 AUSTRALIA
- 14.4.4.1 Robust infrastructure for clinical trials and well-developed healthcare sector to aid market growth
- 14.4.5 SOUTH KOREA
- 14.4.5.1 Growth in biopharmaceutical industry to facilitate market growth
- 14.4.6 REST OF ASIA PACIFIC
- 14.4.1 CHINA
- 14.5 LATIN AMERICA
- 14.5.1 BRAZIL
- 14.5.1.1 Product approvals and high healthcare expenditure to support market growth
- 14.5.2 REST OF LATIN AMERICA
- 14.5.1 BRAZIL
- 14.6 MIDDLE EAST
- 14.6.1 GCC COUNTRIES
- 14.6.2 KINGDOM OF SAUDI ARABIA
- 14.6.2.1 Increased number of product approvals and high healthcare expenditure to boost market growth
- 14.6.3 UAE
- 14.6.3.1 UAE market growth to be driven by localized manufacturing, expanded clinical applications, and strategic collaborations
- 14.6.4 REST OF MIDDLE EAST
- 14.7 AFRICA
- 14.7.1 SIGNIFICANT BURDEN OF HEMATOLOGIC MALIGNANCIES AND INFECTIOUS DISEASES TO PROPEL MARKET GROWTH
15 COMPETITIVE LANDSCAPE
- 15.1 INTRODUCTION
- 15.2 KEY PLAYER STRATEGY/RIGHT TO WIN
- 15.2.1 OVERVIEW OF MAJOR STRATEGIES ADOPTED BY PLAYERS IN CAR T-CELL THERAPY MARKET
- 15.3 REVENUE ANALYSIS, 2023-2025
- 15.4 MARKET SHARE ANALYSIS, 2025
- 15.5 COMPANY EVALUATION MATRIX: KEY PLAYERS, 2025
- 15.5.1 STARS
- 15.5.2 EMERGING LEADERS
- 15.5.3 PERVASIVE PLAYERS
- 15.5.4 PARTICIPANTS
- 15.5.5 COMPANY FOOTPRINT: KEY PLAYERS, 2025
- 15.5.5.1 Company footprint
- 15.5.5.2 Region footprint
- 15.5.5.3 Product footprint
- 15.5.5.4 Target footprint
- 15.5.5.5 Indication footprint
- 15.6 COMPANY EVALUATION MATRIX: STARTUPS/SMES, 2025
- 15.6.1 PROGRESSIVE COMPANIES
- 15.6.2 RESPONSIVE COMPANIES
- 15.6.3 DYNAMIC COMPANIES
- 15.6.4 STARTING BLOCKS
- 15.6.5 COMPETITIVE BENCHMARKING: STARTUPS/SMES, 2025
- 15.6.5.1 Detailed list of key startups/SMEs
- 15.6.5.2 Competitive benchmarking of startups/SMEs
- 15.7 COMPANY VALUATION & FINANCIAL METRICS
- 15.7.1 FINANCIAL METRICS
- 15.7.2 COMPANY VALUATION
- 15.8 BRAND/PRODUCT COMPARISON
- 15.9 COMPETITIVE SCENARIO
- 15.9.1 PRODUCT APPROVALS
- 15.9.2 DEALS
- 15.9.3 EXPANSIONS
- 15.9.4 OTHER DEVELOPMENTS
16 COMPANY PROFILES
- 16.1 KEY PLAYERS
- 16.1.1 JOHNSON & JOHNSON
- 16.1.1.1 Business overview
- 16.1.1.2 Products offered
- 16.1.1.3 Recent developments
- 16.1.1.3.1 Product approvals
- 16.1.1.3.2 Deals
- 16.1.1.4 MnM view
- 16.1.1.4.1 Key strengths
- 16.1.1.4.2 Strategic choices
- 16.1.1.4.3 Weaknesses & competitive threats
- 16.1.2 GILEAD SCIENCES, INC.
- 16.1.2.1 Business overview
- 16.1.2.2 Products offered
- 16.1.2.3 Recent developments
- 16.1.2.3.1 Product approvals
- 16.1.2.3.2 Deals
- 16.1.2.3.3 Other developments
- 16.1.2.4 MnM view
- 16.1.2.4.1 Key strengths
- 16.1.2.4.2 Strategic choices
- 16.1.2.4.3 Weaknesses & competitive threats
- 16.1.3 BRISTOL-MYERS SQUIBB COMPANY
- 16.1.3.1 Business overview
- 16.1.3.2 Products offered
- 16.1.3.3 Recent developments
- 16.1.3.3.1 Product approvals
- 16.1.3.3.2 Deals
- 16.1.3.4 MnM view
- 16.1.3.4.1 Key strengths
- 16.1.3.4.2 Strategic choices
- 16.1.3.4.3 Weaknesses & competitive threats
- 16.1.4 NOVARTIS AG
- 16.1.4.1 Business overview
- 16.1.4.2 Products offered
- 16.1.4.3 Recent developments
- 16.1.4.3.1 Product approvals
- 16.1.4.3.2 Other developments
- 16.1.4.4 MnM view
- 16.1.4.4.1 Key strengths
- 16.1.4.4.2 Strategic choices
- 16.1.4.4.3 Weaknesses & competitive threats
- 16.1.5 AUTOLUS THERAPEUTICS
- 16.1.5.1 Business overview
- 16.1.5.2 Products offered
- 16.1.5.3 Recent developments
- 16.1.5.3.1 Product approvals
- 16.1.5.3.2 Deals
- 16.1.5.4 MnM view
- 16.1.5.4.1 Key strengths
- 16.1.5.4.2 Strategic choices
- 16.1.5.4.3 Weaknesses & competitive threats
- 16.1.6 JW (CAYMAN) THERAPEUTICS CO., LTD.
- 16.1.6.1 Business overview
- 16.1.6.2 Products offered
- 16.1.6.3 Recent developments
- 16.1.6.3.1 Product approvals
- 16.1.6.3.2 Deals
- 16.1.7 IMMUNOADOPTIVE CELL THERAPY PRIVATE LIMITED (IMMUNOACT)
- 16.1.7.1 Business overview
- 16.1.7.2 Products offered
- 16.1.7.3 Recent developments
- 16.1.7.3.1 Deals
- 16.1.7.3.2 Other developments
- 16.1.8 CARSGEN THERAPEUTICS HOLDINGS LIMITED
- 16.1.8.1 Business overview
- 16.1.8.2 Products offered
- 16.1.8.3 Recent developments
- 16.1.8.3.1 Product approvals
- 16.1.8.3.2 Deals
- 16.1.8.3.3 Expansions
- 16.1.9 IASO BIOTHERAPEUTICS
- 16.1.9.1 Business overview
- 16.1.9.2 Products offered
- 16.1.9.3 Recent developments
- 16.1.9.3.1 Product approvals
- 16.1.9.3.2 Deals
- 16.1.10 IMMUNEEL THERAPEUTICS
- 16.1.10.1 Business overview
- 16.1.10.2 Products offered
- 16.1.10.3 Recent developments
- 16.1.10.3.1 Product launches
- 16.1.1 JOHNSON & JOHNSON
- 16.2 OTHER PLAYERS
- 16.2.1 WUGEN
- 16.2.2 CARTESIAN THERAPEUTICS, INC.
- 16.2.3 ALLOGENE THERAPEUTICS
- 16.2.4 LYELL IMMUNOPHARMA, INC.
- 16.2.5 KYVERNA THERAPEUTICS, INC.
- 16.2.6 CELLICTIS SA
- 16.2.7 BRAINCHILD BIO
- 16.2.8 ATARA BIOTHERAPEUTICS, INC.
- 16.2.9 CARIBOU BIOSCIENCES
- 16.2.10 ARCELLX
- 16.2.11 CRISPR THERAPEUTICS
- 16.2.12 POSEIDA THERAPEUTICS
- 16.2.13 CABALETTA BIO
17 RESEARCH METHODOLOGY
- 17.1 RESEARCH DATA
- 17.1.1 SECONDARY DATA
- 17.1.1.1 Objectives of secondary research
- 17.1.2 PRIMARY DATA
- 17.1.2.1 Breakdown of primaries
- 17.1.2.2 Objectives of primary research
- 17.1.1 SECONDARY DATA
- 17.2 MARKET SIZE ESTIMATION
- 17.2.1 BOTTOM-UP APPROACH
- 17.2.2 INSIGHTS FROM PRIMARY EXPERTS
- 17.2.3 SEGMENTAL MARKET SIZE ESTIMATION
- 17.3 GROWTH RATE ASSUMPTIONS
- 17.3.1 CAGR PROJECTIONS
- 17.3.2 IMPACT OF SUPPLY- AND DEMAND-SIDE FACTORS
- 17.4 DATA TRIANGULATION
- 17.5 RESEARCH LIMITATIONS
- 17.6 STUDY ASSUMPTIONS
- 17.7 RISK ANALYSIS
18 APPENDIX
- 18.1 DISCUSSION GUIDE
- 18.2 KNOWLEDGESTORE: MARKETSANDMARKETS' SUBSCRIPTION PORTAL
- 18.3 CUSTOMIZATION OPTIONS
- 18.4 RELATED REPORTS
- 18.5 AUTHOR DETAILS
List of Tables
- TABLE 1 CAR T-CELL THERAPY MARKET: INCLUSIONS & EXCLUSIONS
- TABLE 2 KEY PIPELINE PRODUCTS IN PHASE-2 AND PHASE-3 CAR T-CELL THERAPY MARKET (AS OF 2025)
- TABLE 3 IMPACT OF PORTER'S FIVE FORCES ON CAR T-CELL THERAPY MARKET
- TABLE 4 ROLE OF MAJOR COMPANIES IN CAR T-CELL THERAPY MARKET ECOSYSTEM (SUPPLY AND DEMAND SIDES)
- TABLE 5 AVERAGE SELLING PRICE OF CAR T-CELL THERAPY PRODUCTS, BY TYPE, 2025 (USD)
- TABLE 6 AVERAGE SELLING PRICE OF CAR T-CELL THERAPY PRODUCTS, BY REGION, 2025
- TABLE 7 KEY CONFERENCES & EVENTS IN CAR T-CELL THERAPY MARKET, JANUARY 2026-DECEMBER 2027
- TABLE 8 US-ADJUSTED RECIPROCAL TARIFF RATES
- TABLE 9 NORTH AMERICA: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 10 EUROPE: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 11 ASIA PACIFIC: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 12 REST OF THE WORLD: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 13 COUNTRY-WISE REGULATORY SCENARIO FOR CAR T-CELL THERAPY PRODUCT MANUFACTURERS
- TABLE 14 CAR T-CELL THERAPY MARKET: REIMBURSEMENT STATUS AND FUNDING CHANNEL, BY COUNTRY/REGION (AS OF 2026)
- TABLE 15 CAR T-CELL THERAPY MARKET: HCPCS CODES OF COMMERCIALIZED THERAPIES
- TABLE 16 COMBINATION THERAPY TRIALS IN CAR T-CELL THERAPY MARKET
- TABLE 17 INFLUENCE OF KEY STAKEHOLDERS ON BUYING PROCESS, BY END USER (%)
- TABLE 18 KEY BUYING CRITERIA, BY END USER
- TABLE 19 CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024-2031 (USD MILLION)
- TABLE 20 CAR T-CELL THERAPY MARKET, BY REGION, 2024-2031 (NUMBER OF THERAPIES)
- TABLE 21 CAR T-CELL THERAPY MARKET FOR ABECMA, BY REGION, 2024-2031 (USD MILLION)
- TABLE 22 NORTH AMERICA: CAR T-CELL THERAPY MARKET FOR ABECMA, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 23 EUROPE: CAR T-CELL THERAPY MARKET FOR ABECMA, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 24 ASIA PACIFIC: CAR T-CELL THERAPY MARKET FOR ABECMA, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 25 LATIN AMERICA: CAR T-CELL THERAPY MARKET FOR ABECMA, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 26 MIDDLE EAST: CAR T-CELL THERAPY MARKET FOR ABECMA, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 27 GCC COUNTRIES: CAR T-CELL THERAPY MARKET FOR ABECMA, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 28 CAR T-CELL THERAPY MARKET FOR BREYANZI, BY REGION, 2024-2031 (USD MILLION)
- TABLE 29 NORTH AMERICA: CAR T-CELL THERAPY MARKET FOR BREYANZI, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 30 EUROPE: CAR T-CELL THERAPY MARKET FOR BREYANZI, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 31 ASIA PACIFIC: CAR T-CELL THERAPY MARKET FOR BREYANZI, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 32 LATIN AMERICA: CAR T-CELL THERAPY MARKET FOR BREYANZI, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 33 MIDDLE EAST: CAR T-CELL THERAPY MARKET FOR BREYANZI, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 34 GCC COUNTRIES: CAR T-CELL THERAPY MARKET FOR BREYANZI, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 35 CAR T-CELL THERAPY MARKET FOR CARVYKTI, BY REGION, 2024-2031 (USD MILLION)
- TABLE 36 NORTH AMERICA: CAR T-CELL THERAPY MARKET FOR CARVYKTI, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 37 EUROPE: CAR T-CELL THERAPY MARKET FOR CARVYKTI, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 38 ASIA PACIFIC: CAR T-CELL THERAPY MARKET FOR CARVYKTI, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 39 LATIN AMERICA: CAR T-CELL THERAPY MARKET FOR CARVYKTI, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 40 MIDDLE EAST: CAR T-CELL THERAPY MARKET FOR CARVYKTI, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 41 GCC COUNTRIES: CAR T-CELL THERAPY MARKET FOR CARVYKTI, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 42 CAR T-CELL THERAPY MARKET FOR YESCARTA, BY REGION, 2024-2031 (USD MILLION)
- TABLE 43 NORTH AMERICA: CAR T-CELL THERAPY MARKET FOR YESCARTA, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 44 EUROPE: CAR T-CELL THERAPY MARKET FOR YESCARTA, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 45 ASIA PACIFIC: CAR T-CELL THERAPY MARKET FOR YESCARTA, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 46 LATIN AMERICA: CAR T-CELL THERAPY MARKET FOR YESCARTA, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 47 MIDDLE EAST: CAR T-CELL THERAPY MARKET FOR YESCARTA, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 48 GCC COUNTRIES: CAR T-CELL THERAPY MARKET FOR YESCARTA, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 49 CAR T-CELL THERAPY MARKET FOR TECARTUS, BY REGION, 2024-2031 (USD MILLION)
- TABLE 50 NORTH AMERICA: CAR T-CELL THERAPY MARKET FOR TECARTUS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 51 EUROPE: CAR T-CELL THERAPY MARKET FOR TECARTUS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 52 ASIA PACIFIC: CAR T-CELL THERAPY MARKET FOR TECARTUS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 53 LATIN AMERICA: CAR T-CELL THERAPY MARKET FOR TECARTUS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 54 MIDDLE EAST: CAR T-CELL THERAPY MARKET FOR TECARTUS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 55 GCC COUNTRIES: CAR T-CELL THERAPY MARKET FOR TECARTUS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 56 CAR T-CELL THERAPY MARKET FOR KYMRIAH, BY REGION, 2024-2031 (USD MILLION)
- TABLE 57 NORTH AMERICA: CAR T-CELL THERAPY MARKET FOR KYMRIAH, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 58 EUROPE: CAR T-CELL THERAPY MARKET FOR KYMRIAH, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 59 ASIA PACIFIC: CAR T-CELL THERAPY MARKET FOR KYMRIAH, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 60 LATIN AMERICA: CAR T-CELL THERAPY MARKET FOR KYMRIAH, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 61 MIDDLE EAST: CAR T-CELL THERAPY MARKET FOR KYMRIAH, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 62 GCC COUNTRIES: CAR T-CELL THERAPY MARKET FOR KYMRIAH, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 63 CAR T-CELL THERAPY MARKET FOR OTHER PRODUCTS, BY REGION, 2024-2031 (USD MILLION)
- TABLE 64 NORTH AMERICA: CAR T-CELL THERAPY MARKET FOR OTHER PRODUCTS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 65 EUROPE: CAR T-CELL THERAPY MARKET FOR OTHER PRODUCTS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 66 ASIA PACIFIC: CAR T-CELL THERAPY MARKET FOR OTHER PRODUCTS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 67 LATIN AMERICA: CAR T-CELL THERAPY MARKET FOR OTHER PRODUCTS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 68 MIDDLE EAST: CAR T-CELL THERAPY MARKET FOR OTHER PRODUCTS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 69 GCC COUNTRIES: CAR T-CELL THERAPY MARKET FOR OTHER PRODUCTS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 70 CAR T-CELL THERAPY MARKET, BY TARGET, 2024-2031 (USD MILLION)
- TABLE 71 COMMERCIALLY AVAILABLE CD19 ANTIGEN TARGETING CAR T-CELL THERAPIES
- TABLE 72 CD19-TARGETING CAR T-CELL THERAPY MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 73 NORTH AMERICA: CD19-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 74 EUROPE: CD19-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 75 ASIA PACIFIC: CD19-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 76 LATIN AMERICA: CD19-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 77 MIDDLE EAST: CD19-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 78 GCC COUNTRIES: CD19-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 79 COMMERCIALLY AVAILABLE BCMA ANTIGEN TARGETING CAR T-CELL THERAPIES
- TABLE 80 BCMA-TARGETING CAR T-CELL THERAPY MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 81 NORTH AMERICA: BCMA-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 82 EUROPE: BCMA-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 83 ASIA PACIFIC: BCMA-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 84 LATIN AMERICA: BCMA-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 85 MIDDLE EAST: BCMA-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 86 GCC COUNTRIES: BCMA-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 87 CD19/20-TARGETING CAR T-CELL THERAPY MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 88 NORTH AMERICA: CD19/20-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 89 EUROPE: CD19/20-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 90 ASIA PACIFIC: CD19/20-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 91 LATIN AMERICA: CD19/20-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 92 MIDDLE EAST: CD19/20-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 93 GCC COUNTRIES: CD19/20-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 94 CD7-TARGETING CAR T-CELL THERAPY MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 95 NORTH AMERICA: CD7-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 96 EUROPE: CD7-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 97 ASIA PACIFIC: CD7-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 98 LATIN AMERICA: CD7-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 99 MIDDLE EAST: CD7-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 100 GCC COUNTRIES: CD7-TARGETING CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 101 OTHER CAR T-CELL THERAPY TARGETS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 102 NORTH AMERICA: OTHER CAR T-CELL THERAPY TARGETS MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 103 EUROPE: OTHER CAR T-CELL THERAPY TARGETS MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 104 ASIA PACIFIC: OTHER CAR T-CELL THERAPY TARGETS MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 105 LATIN AMERICA: OTHER CAR T-CELL THERAPY TARGETS MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 106 MIDDLE EAST: OTHER CAR T-CELL THERAPY TARGETS MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 107 GCC COUNTRIES: OTHER CAR T-CELL THERAPY TARGETS MARKETA, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 108 CAR T-CELL THERAPY MARKET, BY INDICATION, 2024-2031 (USD MILLION)
- TABLE 109 CAR T-CELL THERAPY MARKET FOR B-CELL LYMPHOMA (BCL), BY REGION, 2024-2031 (USD MILLION)
- TABLE 110 NORTH AMERICA: CAR T-CELL THERAPY MARKET FOR B-CELL LYMPHOMA (BCL), BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 111 EUROPE: CAR T-CELL THERAPY MARKET FOR B-CELL LYMPHOMA (BCL), BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 112 ASIA PACIFIC: CAR T-CELL THERAPY MARKET FOR B-CELL LYMPHOMA (BCL), BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 113 LATIN AMERICA: CAR T-CELL THERAPY MARKET FOR B-CELL LYMPHOMA (BCL), BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 114 MIDDLE EAST: CAR T-CELL THERAPY MARKET FOR B-CELL LYMPHOMA (BCL), BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 115 GCC COUNTRIES: CAR T-CELL THERAPY MARKET FOR B-CELL LYMPHOMA (BCL), BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 116 CAR T-CELL THERAPY MARKET FOR MULTIPLE MYELOMA, BY REGION, 2024-2031(USD MILLION)
- TABLE 117 NORTH AMERICA: CAR T-CELL THERAPY MARKET FOR MULTIPLE MYELOMA, BY COUNTRY, 2024-2031(USD MILLION)
- TABLE 118 EUROPE: CAR T-CELL THERAPY MARKET FOR MULTIPLE MYELOMA, BY COUNTRY, 2024-2031(USD MILLION)
- TABLE 119 ASIA PACIFIC: CAR T-CELL THERAPY MARKET FOR MULTIPLE MYELOMA, BY COUNTRY, 2024-2031(USD MILLION)
- TABLE 120 LATIN AMERICA: CAR T-CELL THERAPY MARKET FOR MULTIPLE MYELOMA, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 121 MIDDLE EAST: CAR T-CELL THERAPY MARKET FOR MULTIPLE MYELOMA, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 122 GCC COUNTRIES: CAR T-CELL THERAPY MARKET FOR MULTIPLE MYELLOMA, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 123 CAR T-CELL THERAPY MARKET FOR ACUTE LYMPHOBLASTIC LEUKEMIA (ALL), BY REGION, 2024-2031(USD MILLION)
- TABLE 124 NORTH AMERICA: CAR T-CELL THERAPY MARKET FOR ACUTE LYMPHOBLASTIC LEUKEMIA (ALL), BY COUNTRY, 2024-2031(USD MILLION)
- TABLE 125 EUROPE: CAR T-CELL THERAPY MARKET FOR ACUTE LYMPHOBLASTIC LEUKEMIA (ALL), BY COUNTRY, 2024-2031(USD MILLION)
- TABLE 126 ASIA PACIFIC: CAR T-CELL THERAPY MARKET FOR ACUTE LYMPHOBLASTIC LEUKEMIA (ALL), BY COUNTRY, 2024-2031(USD MILLION)
- TABLE 127 LATIN AMERICA: CAR T-CELL THERAPY MARKET FOR ACUTE LYMPHOBLASTIC LEUKEMIA (ALL), BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 128 MIDDLE EAST: CAR T-CELL THERAPY MARKET FOR ACUTE LYMPHOBLASTIC LEUKEMIA (ALL), BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 129 GCC COUNTRIES: CAR T-CELL THERAPY MARKET FOR ACUTE LYMPHOBLASTIC LEUKEMIA (ALL), BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 130 CAR T-CELL THERAPY MARKET FOR OTHER INDICATIONS, BY REGION, 2024-2031 (USD MILLION)
- TABLE 131 NORTH AMERICA: CAR T-CELL THERAPY MARKET FOR OTHER INDICATIONS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 132 EUROPE: CAR T-CELL THERAPY MARKET FOR OTHER INDICATIONS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 133 ASIA PACIFIC: CAR T-CELL THERAPY MARKET FOR OTHER INDICATIONS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 134 LATIN AMERICA: CAR T-CELL THERAPY MARKET FOR OTHER INDICATIONS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 135 MIDDLE EAST: CAR T-CELL THERAPY MARKET FOR OTHER INDICATIONS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 136 GCC COUNTRIES: CAR T-CELL THERAPY MARKET FOR OTHER INDICATIONS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 137 CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024-2031 (USD MILLION)
- TABLE 138 CAR T-CELL THERAPY MARKET FOR ADULTS, BY REGION, 2024-2031 (USD MILLION)
- TABLE 139 NORTH AMERICA: CAR T-CELL THERAPY MARKET FOR ADULTS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 140 EUROPE: CAR T-CELL THERAPY MARKET FOR ADULTS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 141 ASIA PACIFIC: CAR T-CELL THERAPY MARKET FOR ADULTS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 142 LATIN AMERICA: CAR T-CELL THERAPY MARKET FOR ADULTS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 143 MIDDLE EAST: CAR T-CELL THERAPY MARKET FOR ADULTS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 144 GCC COUNTRIES: CAR T-CELL THERAPY MARKET FOR ADULTS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 145 CAR T-CELL THERAPY MARKET FOR PEDIATRIC, BY REGION, 2024-2031 (USD MILLION)
- TABLE 146 NORTH AMERICA: CAR T-CELL THERAPY MARKET FOR PEDIATRIC, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 147 EUROPE: CAR T-CELL THERAPY MARKET FOR PEDIATRIC, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 148 ASIA PACIFIC: CAR T-CELL THERAPY MARKET FOR PEDIATRIC, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 149 LATIN AMERICA: CAR T-CELL THERAPY MARKET FOR PEDIATRIC, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 150 MIDDLE EAST: CAR T-CELL THERAPY MARKET FOR PEDIATRIC, BY COUNTRY, 2024-2031(USD MILLION)
- TABLE 151 GCC COUNTRIES: CAR T-CELL THERAPY MARKET FOR PEDIATRIC, BY COUNTRY, 2024-2031(USD MILLION)
- TABLE 152 CAR T-CELL THERAPY MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 153 CAR T-CELL THERAPY MARKET FOR HOSPITALS, BY REGION, 2024-2031 (USD MILLION)
- TABLE 154 NORTH AMERICA: CAR T-CELL THERAPY MARKET FOR HOSPITALS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 155 EUROPE: CAR T-CELL THERAPY MARKET FOR HOSPITALS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 156 ASIA PACIFIC: CAR T-CELL THERAPY MARKET FOR HOSPITALS, BY COUNTRY, 2024-2031(USD MILLION)
- TABLE 157 LATIN AMERICA: CAR T-CELL THERAPY MARKET FOR HOSPITALS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 158 MIDDLE EAST: CAR T-CELL THERAPY MARKET FOR HOSPITALS, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 159 GCC COUNTRIES: CAR T-CELL THERAPY MARKET FOR HOSPITALS, BY COUNTRY, 2024-2031(USD MILLION)
- TABLE 160 CAR T-CELL THERAPY MARKET FOR SPECIALTY CENTRES, BY REGION, 2024-2031 (USD MILLION)
- TABLE 161 NORTH AMERICA: CAR T-CELL THERAPY MARKET FOR SPECIALTY CENTRES, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 162 EUROPE: CAR T-CELL THERAPY MARKET FOR SPECIALTY CENTRES, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 163 ASIA PACIFIC: CAR T-CELL THERAPY MARKET FOR SPECIALTY CENTRES, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 164 LATIN AMERICA: CAR T-CELL THERAPY MARKET FOR SPECIALTY CENTRES, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 165 MIDDLE EAST: CAR T-CELL THERAPY MARKET FOR SPECIALTY CENTRES, BY COUNTRY, 2024-2031(USD MILLION)
- TABLE 166 GCC COUNTRIES: CAR T-CELL THERAPY MARKET FOR SPECIALTY CENTRES, BY COUNTRY, 2024-2031(USD MILLION)
- TABLE 167 CAR T-CELL THERAPY MARKET FOR LONG-TERM CARE FACILITIES, BY REGION, 2024-2031(USD MILLION)
- TABLE 168 NORTH AMERICA: CAR T-CELL THERAPY MARKET FOR LONG-TERM CARE FACILITIES, BY COUNTRY, 2024-2031(USD MILLION)
- TABLE 169 EUROPE: CAR T-CELL THERAPY MARKET FOR LONG-TERM CARE FACILITIES, BY COUNTRY, 2024-2031(USD MILLION)
- TABLE 170 ASIA PACIFIC: CAR T-CELL THERAPY MARKET FOR LONG-TERM CARE FACILITIES, BY COUNTRY, 2024-2031(USD MILLION)
- TABLE 171 LATIN AMERICA: CAR T-CELL THERAPY MARKET FOR LONG-TERM CARE FACILITIES, BY COUNTRY, 2024-2031(USD MILLION)
- TABLE 172 MIDDLE EAST: CAR T-CELL THERAPY MARKET FOR LONG-TERM CARE FACILITIES, BY COUNTRY, 2024-2031(USD MILLION)
- TABLE 173 GCC COUNTRIES: CAR T-CELL THERAPY MARKET FOR LONG-TERM CARE FACILITIES, BY COUNTRY, 2024-2031(USD MILLION)
- TABLE 174 CAR T-CELL THERAPY MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 175 NORTH AMERICA: CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 176 NORTH AMERICA: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024-2031 (USD MILLION)
- TABLE 177 NORTH AMERICA: CAR T-CELL THERAPY MARKET, BY TARGET, 2024-2031 (USD MILLION)
- TABLE 178 NORTH AMERICA: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024-2031 (USD MILLION)
- TABLE 179 NORTH AMERICA: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024-2031 (USD MILLION)
- TABLE 180 NORTH AMERICA: CAR T-CELL THERAPY MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 181 US: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024-2031 (USD MILLION)
- TABLE 182 US: CAR T-CELL THERAPY MARKET, BY TARGET, 2024-2031 (USD MILLION)
- TABLE 183 US: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024-2031 (USD MILLION)
- TABLE 184 US: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024-2031 (USD MILLION)
- TABLE 185 US: CAR T-CELL THERAPY MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 186 CANADA: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024-2031 (USD MILLION)
- TABLE 187 CANADA: CAR T-CELL THERAPY MARKET, BY TARGET, 2024-2031 (USD MILLION)
- TABLE 188 CANADA: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024-2031 (USD MILLION)
- TABLE 189 CANADA: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024-2031 (USD MILLION)
- TABLE 190 CANADA: CAR T-CELL THERAPY MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 191 EUROPE: CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 192 EUROPE: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024-2031 (USD MILLION)
- TABLE 193 EUROPE: CAR T-CELL THERAPY MARKET, BY TARGET, 2024-2031 (USD MILLION)
- TABLE 194 EUROPE: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024-2031 (USD MILLION)
- TABLE 195 EUROPE: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024-2031 (USD MILLION)
- TABLE 196 EUROPE: CAR T-CELL THERAPY MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 197 GERMANY: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024-2031 (USD MILLION)
- TABLE 198 GERMANY: CAR T-CELL THERAPY MARKET, BY TARGET, 2024-2031 (USD MILLION)
- TABLE 199 GERMANY: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024-2031 (USD MILLION)
- TABLE 200 GERMANY: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024-2031 (USD MILLION)
- TABLE 201 GERMANY: CAR T-CELL THERAPY MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 202 UK: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024-2031 (USD MILLION)
- TABLE 203 UK: CAR T-CELL THERAPY MARKET, BY TARGET, 2024-2031 (USD MILLION)
- TABLE 204 UK: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024-2031 (USD MILLION)
- TABLE 205 UK: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024-2031 (USD MILLION)
- TABLE 206 UK: CAR T-CELL THERAPY MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 207 FRANCE: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024-2031 (USD MILLION)
- TABLE 208 FRANCE: CAR T-CELL THERAPY MARKET, BY TARGET, 2024-2031 (USD MILLION)
- TABLE 209 FRANCE: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024-2031 (USD MILLION)
- TABLE 210 FRANCE: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024-2031 (USD MILLION)
- TABLE 211 FRANCE: CAR T-CELL THERAPY MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 212 ITALY: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024-2031 (USD MILLION)
- TABLE 213 ITALY: CAR T-CELL THERAPY MARKET, BY TARGET, 2024-2031 (USD MILLION)
- TABLE 214 ITALY: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024-2031 (USD MILLION)
- TABLE 215 ITALY: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024-2031 (USD MILLION)
- TABLE 216 ITALY: CAR T-CELL THERAPY MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 217 SPAIN: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024-2031 (USD MILLION)
- TABLE 218 SPAIN: CAR T-CELL THERAPY MARKET, BY TARGET, 2024-2031 (USD MILLION)
- TABLE 219 SPAIN: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024-2031 (USD MILLION)
- TABLE 220 SPAIN: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024-2031 (USD MILLION)
- TABLE 221 SPAIN: CAR T-CELL THERAPY MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 222 REST OF EUROPE: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024-2031 (USD MILLION)
- TABLE 223 REST OF EUROPE: CAR T-CELL THERAPY MARKET, BY TARGET, 2024-2031 (USD MILLION)
- TABLE 224 REST OF EUROPE: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024-2031 (USD MILLION)
- TABLE 225 REST OF EUROPE: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024-2031 (USD MILLION)
- TABLE 226 REST OF EUROPE: CAR T-CELL THERAPY MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 227 ASIA PACIFIC: CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 228 ASIA PACIFIC: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024-2031 (USD MILLION)
- TABLE 229 ASIA PACIFIC: CAR T-CELL THERAPY MARKET, BY TARGET, 2024-2031 (USD MILLION)
- TABLE 230 ASIA PACIFIC: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024-2031 (USD MILLION)
- TABLE 231 ASIA PACIFIC: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024-2031 (USD MILLION)
- TABLE 232 ASIA PACIFIC: CAR T-CELL THERAPY MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 233 CHINA: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024-2031 (USD MILLION)
- TABLE 234 CHINA: CAR T-CELL THERAPY MARKET, BY TARGET, 2024-2031 (USD MILLION)
- TABLE 235 CHINA: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024-2031 (USD MILLION)
- TABLE 236 CHINA: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024-2031 (USD MILLION)
- TABLE 237 CHINA: CAR T-CELL THERAPY MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 238 JAPAN: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024-2031 (USD MILLION)
- TABLE 239 JAPAN: CAR T-CELL THERAPY MARKET, BY TARGET, 2024-2031 (USD MILLION)
- TABLE 240 JAPAN: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024-2031 (USD MILLION)
- TABLE 241 JAPAN: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024-2031 (USD MILLION)
- TABLE 242 JAPAN: CAR T-CELL THERAPY MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 243 INDIA: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024-2031 (USD MILLION)
- TABLE 244 INDIA: CAR T-CELL THERAPY MARKET, BY TARGET, 2024-2031 (USD MILLION)
- TABLE 245 INDIA: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024-2031 (USD MILLION)
- TABLE 246 INDIA: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024-2031 (USD MILLION)
- TABLE 247 INDIA: CAR T-CELL THERAPY MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 248 AUSTRALIA: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024-2031 (USD MILLION)
- TABLE 249 AUSTRALIA: CAR T-CELL THERAPY MARKET, BY TARGET, 2024-2031 (USD MILLION)
- TABLE 250 AUSTRALIA: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024-2031 (USD MILLION)
- TABLE 251 AUSTRALIA: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024-2031 (USD MILLION)
- TABLE 252 AUSTRALIA: CAR T-CELL THERAPY MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 253 SOUTH KOREA: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024-2031 (USD MILLION)
- TABLE 254 SOUTH KOREA: CAR T-CELL THERAPY MARKET, BY TARGET, 2024-2031 (USD MILLION)
- TABLE 255 SOUTH KOREA: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024-2031 (USD MILLION)
- TABLE 256 SOUTH KOREA: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024-2031 (USD MILLION)
- TABLE 257 SOUTH KOREA: CAR T-CELL THERAPY MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 258 REST OF ASIA PACIFC: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024-2031 (USD MILLION)
- TABLE 259 REST OF ASIA PACIFIC: CAR T-CELL THERAPY MARKET, BY TARGET, 2024-2031 (USD MILLION)
- TABLE 260 REST OF ASIA PACIFIC: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024-2031 (USD MILLION)
- TABLE 261 REST OF ASIA PACIFIC: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024-2031 (USD MILLION)
- TABLE 262 REST OF ASIA PACIFIC: CAR T-CELL THERAPY MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 263 LATIN AMERICA: CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 264 LATIN AMERICA: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024-2031 (USD MILLION)
- TABLE 265 LATIN AMERICA: CAR T-CELL THERAPY MARKET, BY TARGET, 2024-2031 (USD MILLION)
- TABLE 266 LATIN AMERICA: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024-2031 (USD MILLION)
- TABLE 267 LATIN AMERICA: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024-2031 (USD MILLION)
- TABLE 268 LATIN AMERICA: CAR T-CELL THERAPY MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 269 BRAZIL: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024-2031 (USD MILLION)
- TABLE 270 BRAZIL: CAR T-CELL THERAPY MARKET, BY TARGET, 2024-2031 (USD MILLION)
- TABLE 271 BRAZIL: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024-2031 (USD MILLION)
- TABLE 272 BRAZIL: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024-2031 (USD MILLION)
- TABLE 273 BRAZIL: CAR T-CELL THERAPY MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 274 REST OF LATIN AMERICA: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024-2031 (USD MILLION)
- TABLE 275 REST OF LATIN AMERICA: CAR T-CELL THERAPY MARKET, BY TARGET, 2024-2031 (USD MILLION)
- TABLE 276 REST OF LATIN AMERICA: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024-2031 (USD MILLION)
- TABLE 277 REST OF LATIN AMERICA: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024-2031 (USD MILLION)
- TABLE 278 REST OF LATIN AMERICA: CAR T-CELL THERAPY MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 279 MIDDLE EAST: CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 280 MIDDLE EAST: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024-2031 (USD MILLION)
- TABLE 281 MIDDLE EAST: CAR T-CELL THERAPY MARKET, BY TARGET, 2024-2031 (USD MILLION)
- TABLE 282 MIDDLE EAST: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024-2031 (USD MILLION)
- TABLE 283 MIDDLE EAST: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024-2031 (USD MILLION)
- TABLE 284 MIDDLE EAST: CAR T-CELL THERAPY MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 285 GCC COUNTRIES: CAR T-CELL THERAPY MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 286 GCC COUNTRIES: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024-2031 (USD MILLION)
- TABLE 287 GCC COUNTRIES: CAR T-CELL THERAPY MARKET, BY TARGET, 2024-2031 (USD MILLION)
- TABLE 288 GCC COUNTRIES: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024-2031 (USD MILLION)
- TABLE 289 GCC COUNTRIES: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024-2031 (USD MILLION)
- TABLE 290 GCC COUNTRIES: CAR T-CELL THERAPY MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 291 KINGDOM OF SAUDI ARABIA: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024-2031 (USD MILLION)
- TABLE 292 KINGDOM OF SAUDI ARABIA: CAR T-CELL THERAPY MARKET, BY TARGET, 2024-2031 (USD MILLION)
- TABLE 293 KINGDOM OF SAUDI ARABIA: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024-2031 (USD MILLION)
- TABLE 294 KINGDOM OF SAUDI ARABIA: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024-2031 (USD MILLION)
- TABLE 295 KINGDOM OF SAUDI ARABIA: CAR T-CELL THERAPY MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 296 UAE: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024-2031 (USD MILLION)
- TABLE 297 UAE: CAR T-CELL THERAPY MARKET, BY TARGET, 2024-2031 (USD MILLION)
- TABLE 298 UAE: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024-2031 (USD MILLION)
- TABLE 299 UAE: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024-2031 (USD MILLION)
- TABLE 300 UAE: CAR T-CELL THERAPY MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 301 REST OF MIDDLE EAST: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024-2031 (USD MILLION)
- TABLE 302 REST OF MIDDLE EAST: CAR T-CELL THERAPY MARKET, BY TARGET, 2024-2031 (USD MILLION)
- TABLE 303 REST OF MIDDLE EAST: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024-2031 (USD MILLION)
- TABLE 304 REST OF MIDDLE EAST: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024-2031 (USD MILLION)
- TABLE 305 REST OF MIDDLE EAST: CAR T-CELL THERAPY MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 306 AFRICA: CAR T-CELL THERAPY MARKET, BY PRODUCT, 2024-2031 (USD MILLION)
- TABLE 307 AFRICA: CAR T-CELL THERAPY MARKET, BY TARGET, 2024-2031 (USD MILLION)
- TABLE 308 AFRICA: CAR T-CELL THERAPY MARKET, BY INDICATION, 2024-2031 (USD MILLION)
- TABLE 309 AFRICA: CAR T-CELL THERAPY MARKET, BY DEMOGRAPHIC, 2024-2031 (USD MILLION)
- TABLE 310 AFRICA: CAR T-CELL THERAPY MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 311 OVERVIEW OF MAJOR STRATEGIES ADOPTED BY KEY PLAYERS IN CAR T-CELL THERAPY MARKET
- TABLE 312 CAR T-CELL THERAPY MARKET: DEGREE OF COMPETITION
- TABLE 313 CAR T-CELL THERAPY MARKET: REGION FOOTPRINT
- TABLE 314 CAR T-CELL THERAPY MARKET: PRODUCT FOOTPRINT
- TABLE 315 CAR T-CELL THERAPY MARKET: TARGET FOOTPRINT
- TABLE 316 CAR T-CELL THERAPY MARKET: INDICATION FOOTPRINT
- TABLE 317 CAR T-CELL THERAPY MARKET: DETAILED LIST OF KEY STARTUPS/SME PLAYERS
- TABLE 318 CAR T-CELL THERAPY MARKET: COMPETITIVE BENCHMARKING OF STARTUPS/SME PLAYERS, BY DEMOGRAPHY AND REGION, 2025
- TABLE 319 CAR T-CELL THERAPY MARKET: PRODUCT APPROVALS, JANUARY 2022-JANUARY 2026
- TABLE 320 CAR T-CELL THERAPY MARKET: DEALS, JANUARY 2022-JANUARY 2026
- TABLE 321 CAR T-CELL THERAPY MARKET: EXPANSIONS, JANUARY 2022-JANUARY 2026
- TABLE 322 CAR T-CELL THERAPY MARKET: OTHER DEVELOPMENTS, JANUARY 2022-JANUARY 2026
- TABLE 323 JOHNSON & JOHNSON: COMPANY OVERVIEW
- TABLE 324 JOHNSON & JOHNSON: PRODUCTS OFFERED
- TABLE 325 JOHNSON & JOHNSON: PRODUCT APPROVALS, JANUARY 2022-JANUARY 2026
- TABLE 326 JOHNSON & JOHNSON: DEALS, JANUARY 2022-JANUARY 2026
- TABLE 327 GILEAD SCIENCES, INC.: COMPANY OVERVIEW
- TABLE 328 GILEAD SCIENCES, INC.: PRODUCTS OFFERED
- TABLE 329 GILEAD SCIENCES, INC.: PRODUCT APPROVALS, JANUARY 2022-JANUARY 2026
- TABLE 330 GILEAD SCIENCES, INC.: DEALS, JANUARY 2022-JANUARY 2026
- TABLE 331 GILEAD SCIENCES, INC.: OTHER DEVELOPMENTS, JANUARY 2022-JANUARY 2026
- TABLE 332 BRISTOL-MYERS SQUIBB COMPANY: COMPANY OVERVIEW
- TABLE 333 BRISTOL-MYERS SQUIBB COMPANY: PRODUCTS OFFERED
- TABLE 334 BRISTOL-MYERS SQUIBB COMPANY: PRODUCT APPROVALS, JANUARY 2022-JANUARY 2026
- TABLE 335 BRISTOL-MYERS SQUIBB COMPANY: DEALS, JANUARY 2022-JANUARY 2026
- TABLE 336 NOVARTIS AG: COMPANY OVERVIEW
- TABLE 337 NOVARTIS AG: PRODUCTS OFFERED
- TABLE 338 NOVARTIS AG: PRODUCT APPROVALS, JANUARY 2022-JANUARY 2026
- TABLE 339 NOVARTIS AG: OTHER DEVELOPMENTS, JANUARY 2022-JANUARY 2026
- TABLE 340 AUTOLUS THERAPEUTICS: COMPANY OVERVIEW
- TABLE 341 AUTOLUS THERAPEUTICS: PRODUCTS OFFERED
- TABLE 342 AUTOLUS THERAPEUTICS: PRODUCT APPROVALS, JANUARY 2022-JANUARY 2026
- TABLE 343 AUTOLUS THERAPEUTICS: DEALS, JANUARY 2022-JANUARY 2026
- TABLE 344 JW (CAYMAN) THERAPEUTICS CO., LTD.: COMPANY OVERVIEW
- TABLE 345 JW (CAYMAN) THERAPEUTICS CO., LTD.: PRODUCTS OFFERED
- TABLE 346 JW (CAYMAN) THERAPEUTICS CO., LTD.: PRODUCT APPROVALS, JANUARY 2022-JANUARY 2026
- TABLE 347 JW (CAYMAN) THERAPEUTICS CO., LTD.: DEALS, JANUARY 2022-JANUARY 2026
- TABLE 348 IMMUNOADOPTIVE CELL THERAPY PRIVATE LIMITED (IMMUNOACT): COMPANY OVERVIEW
- TABLE 349 IMMUNOADOPTIVE CELL THERAPY PRIVATE LIMITED (IMMUNOACT): PRODUCTS OFFERED
- TABLE 350 IMMUNOADOPTIVE CELL THERAPY PRIVATE LIMITED (IMMUNOACT): DEALS, JANUARY 2022-JANUARY 2026
- TABLE 351 IMMUNOADOPTIVE CELL THERAPY PRIVATE LIMITED (IMMUNOACT): OTHER DEVELOPMENTS, JANUARY 2022-JANUARY 2026
- TABLE 352 CARSGEN THERAPEUTICS HOLDINGS LIMITED: COMPANY OVERVIEW
- TABLE 353 CARSGEN THERAPEUTICS HOLDINGS LIMITED: PRODUCTS OFFERED
- TABLE 354 CARSGEN THERAPEUTICS HOLDINGS LIMITED: PRODUCT APPROVALS, JANUARY 2022-JANUARY 2026
- TABLE 355 CARSGEN THERAPEUTICS HOLDINGS LIMITED: DEALS, JANUARY 2022-JANUARY 2026
- TABLE 356 CARSGEN THERAPEUTICS HOLDINGS LIMITED: EXPANSIONS, JANUARY 2022-JANUARY 2026
- TABLE 357 IASO BIOTHERAPEUTICS: COMPANY OVERVIEW
- TABLE 358 IASO BIOTHERAPEUTICS: PRODUCTS OFFERED
- TABLE 359 IASO BIOTHERAPEUTICS: PRODUCT APPROVALS, JANUARY 2022-JANUARY 2026
- TABLE 360 IASO BIOTHERAPEUTICS: DEALS, JANUARY 2022-JANUARY 2026
- TABLE 361 IMMUNEEL THERAPEUTICS: COMPANY OVERVIEW
- TABLE 362 IMMUNEEL THERAPEUTICS: PRODUCTS OFFERED
- TABLE 363 IMMUNEEL THERAPEUTICS: PRODUCT LAUNCHES, JANUARY 2022-JANUARY 2026
- TABLE 364 WUGEN: COMPANY OVERVIEW
- TABLE 365 CARTESIAN THERAPEUTICS, INC.: COMPANY OVERVIEW
- TABLE 366 ALLOGENE THERAPEUTICS: COMPANY OVERVIEW
- TABLE 367 LYELL IMMUNOPHARMA, INC.: COMPANY OVERVIEW
- TABLE 368 KYVERNA THERAPEUTICS, INC.: COMPANY OVERVIEW
- TABLE 369 CELLICTIS SA: COMPANY OVERVIEW
- TABLE 370 BRAINCHILD BIO: COMPANY OVERVIEW
- TABLE 371 ATARA BIOTHERAPEUTICS, INC.: COMPANY OVERVIEW
- TABLE 372 CARIBOU BIOSCIENCES: COMPANY OVERVIEW
- TABLE 373 ARCELLX: COMPANY OVERVIEW
- TABLE 374 CRISPR THERAPEUTICS: COMPANY OVERVIEW
- TABLE 375 POSEIDA THERAPEUTICS: COMPANY OVERVIEW
- TABLE 376 CABALETTA BIO: COMPANY OVERVIEW
- TABLE 377 IMPACT ANALYSIS OF SUPPLY- AND DEMAND-SIDE FACTORS ON CAR T-CELL THERAPY MARKET
- TABLE 378 CAR T-CELL THERAPY MARKET: RISK ANALYSIS
List of Figures
- FIGURE 1 CAR T-CELL THERAPY MARKET SEGMENTATION & REGIONAL SCOPE
- FIGURE 2 CAR T-CELL THERAPY MARKET: YEARS CONSIDERED
- FIGURE 3 CAR T-CELL THERAPY MARKET SCENARIO
- FIGURE 4 GLOBAL CAR T-CELL THERAPY MARKET SIZE, 2024-2031 (USD MILLION)
- FIGURE 5 MAJOR STRATEGIES ADOPTED BY KEY PLAYERS IN CAR T-CELL THERAPY MARKET, 2023-2025
- FIGURE 6 DISRUPTIONS INFLUENCING GROWTH OF CAR T-CELL THERAPY MARKET
- FIGURE 7 HIGH-GROWTH SEGMENTS IN CAR T-CELL THERAPY MARKET, BY PRODUCT, TARGET, END USER, AND REGION, 2026-2031
- FIGURE 8 ASIA PACIFIC TO REGISTER HIGHEST CAGR IN CAR T-CELL THERAPY MARKET, IN TERMS OF VALUE, DURING FORECAST PERIOD
- FIGURE 9 TECHNOLOGICAL ADVANCEMENTS IN CAR T-CELL THERAPIES TO DRIVE GROWTH
- FIGURE 10 US AND ADULTS DOMINATED NORTH AMERICAN CAR T-CELL THERAPY MARKET IN 2025
- FIGURE 11 INDIA TO REGISTER FASTEST GROWTH RATE DURING FORECAST PERIOD
- FIGURE 12 CAR T-CELL THERAPY MARKET: DRIVERS, RESTRAINTS, OPPORTUNITIES, AND CHALLENGES
- FIGURE 13 GLOBAL CAR T-CELL THERAPY TRIALS DISTRIBUTION, BY TARGET (ACROSS ALL PHASES OF DEVELOPMENT), 2025
- FIGURE 14 GLOBAL CAR T-CELL THERAPY TRIALS DISTRIBUTION OF TARGET BY DEVELOPMENT PHASE, 2025
- FIGURE 15 CAR T-CELL THERAPY MARKET: PORTER'S FIVE FORCES ANALYSIS
- FIGURE 16 CAR T-CELL THERAPY MARKET: VALUE CHAIN ANALYSIS
- FIGURE 17 CAR T-CELL THERAPY MARKET: ECOSYSTEM ANALYSIS
- FIGURE 18 AVERAGE SELLING PRICE OF CAR T-CELL THERAPY PRODUCTS, BY KEY PLAYER, 2025 (USD)
- FIGURE 19 TRENDS/DISRUPTIONS IMPACTING CUSTOMER'S BUSINESS IN CAR T-CELL THERAPY MARKET
- FIGURE 20 FUNDING AND NUMBER OF DEALS IN CAR T-CELL THERAPY MARKET (USD MILLION)
- FIGURE 21 IMPACT OF AI/GEN AI ON CAR T-CELL THERAPY MARKET
- FIGURE 22 INFLUENCE OF KEY STAKEHOLDERS ON BUYING PROCESS, BY END USER
- FIGURE 23 KEY BUYING CRITERIA FOR MAJOR END USERS
- FIGURE 24 NORTH AMERICA: CAR T-CELL THERAPY MARKET SNAPSHOT
- FIGURE 25 ASIA PACIFIC: CAR T-CELL THERAPY MARKET SNAPSHOT
- FIGURE 26 REVENUE ANALYSIS OF KEY PLAYERS IN CAR T-CELL THERAPY MARKET, 2023-2025 (USD MILLION)
- FIGURE 27 MARKET SHARE ANALYSIS OF KEY PLAYERS IN CAR T-CELL THERAPY MARKET (2025)
- FIGURE 28 CAR T-CELL THERAPY MARKET: COMPANY EVALUATION MATRIX (KEY PLAYERS), 2025
- FIGURE 29 CAR T-CELL MARKET: COMPANY FOOTPRINT
- FIGURE 30 CAR T-CELL THERAPY MARKET: COMPANY EVALUATION MATRIX (STARTUPS/SMES), 2025
- FIGURE 31 EV/EBITDA OF KEY VENDORS
- FIGURE 32 YEAR-TO-DATE (YTD) PRICE TOTAL RETURN AND 5-YEAR STOCK BETA OF KEY VENDORS
- FIGURE 33 CAR T-CELL THERAPY MARKET: BRAND/PRODUCT COMPARATIVE ANALYSIS
- FIGURE 34 JOHNSON & JOHNSON: COMPANY SNAPSHOT
- FIGURE 35 GILEAD SCIENCES, INC.: COMPANY SNAPSHOT
- FIGURE 36 BRISTOL-MYERS SQUIBB COMPANY: COMPANY SNAPSHOT
- FIGURE 37 NOVARTIS AG: COMPANY SNAPSHOT
- FIGURE 38 JW (CAYMAN) THERAPEUTICS CO., LTD.: COMPANY SNAPSHOT
- FIGURE 39 CARSGEN THERAPEUTICS HOLDINGS LIMITED: COMPANY SNAPSHOT
- FIGURE 40 CAR T-CELL THERAPY MARKET: RESEARCH DESIGN
- FIGURE 41 CAR T-CELL THERAPY MARKET: BREAKDOWN OF DEMAND- AND SUPPLY-SIDE PRIMARIES
- FIGURE 42 CAR T-CELL THERAPY MARKET SIZE ESTIMATION (SUPPLY-SIDE ANALYSIS), 2025
- FIGURE 43 CAR T-CELL THERAPY MARKET SIZE ESTIMATION: COMPANY REVENUE ANALYSIS-BASED ESTIMATION (PRODUCTS), 2025
- FIGURE 44 ILLUSTRATIVE EXAMPLE OF GILEAD SCIENCES, INC.: REVENUE SHARE ANALYSIS (2025)
- FIGURE 45 CAR T-CELL THERAPY MARKET VALIDATION FROM PRIMARY SOURCES
- FIGURE 46 CAR T-CELL THERAPY MARKET SIZE ESTIMATION METHODOLOGY: TOP-DOWN APPROACH
- FIGURE 47 CAR T-CELL THERAPY MARKET: CAGR PROJECTIONS, 2026-2031
- FIGURE 48 CAR T-CELL THERAPY MARKET: DATA TRIANGULATION METHODOLOGY










