 |
市場調查報告書
商品編碼
1956047
全球製備和工藝層析法市場(按產品類型、應用、最終用戶和地區分類)-預測至2031年Preparative and Process Chromatography Market by Product (Preparative, Process ), Application, End User, Region - Global Forecast to 2031 |
||||||
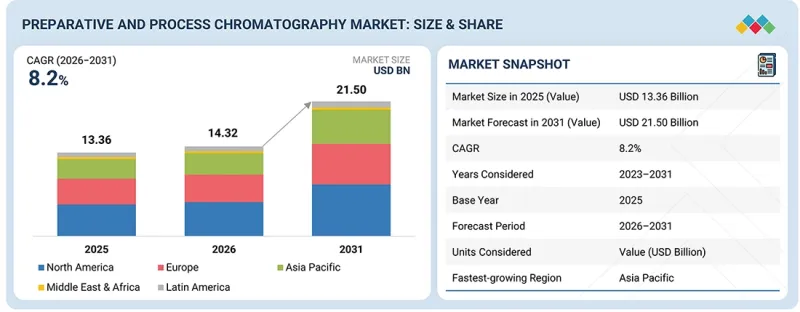
全球製備和工藝層析法市場預計將從 2026 年的 143.2 億美元成長到 2031 年的 215 億美元,預測期內複合年成長率為 8.2%。
| 調查範圍 | |
|---|---|
| 調查期 | 2024-2031 |
| 基準年 | 2025 |
| 預測期 | 2025-2031 |
| 目標單元 | 金額(十億美元) |
| 部分 | 按產品類型、應用程式、最終用戶和地區分類 |
| 目標區域 | 北美、歐洲、亞太地區、拉丁美洲、中東和非洲 |
隨著一些已開發國家對個人化治療的需求不斷成長,採用CGT、寡核苷酸和mRNA等新型技術生產的疫苗和重組蛋白的需求預計將大幅增加。這種需求的成長將需要對這些種類繁多的生物製藥進行純化。這種轉變使得層析法成為一項關鍵的單元操作,尤其是在捕獲和純化方面,從而為市場成長創造了空間。全球單株抗體(mAb)產品線正在不斷擴展,特別是隨著FDA核准數量的增加以及新型雙特異性抗體和抗體藥物複合體(ADC)的上市。這推動了終端用戶對純化需求的成長,預計將帶動製備型和製程型層析法產品的市場發展。
根據產品類型,製備色譜和工藝層析法市場可分為製備層析法和工藝層析法。工藝層析法佔據了最大的市場佔有率。包括大型製藥企業和合約研發生產機構(CDMO)在內的全球製藥和生物製藥終端用戶,正將目光聚焦於專為製程層析法而非製備階段設計的商業化下游工作流程。新建工廠和大型生產設施推動了對製程樹脂、色譜柱及相關服務的持續多年需求。這些因素使得製程規模的層析法產品佔據了全球市場最大的佔有率。
按應用領域分類,製備和工藝層析法市場可細分為單株抗體和重組蛋白、疫苗、胜肽和寡核苷酸、細胞和基因治療以及其他領域。預計到2025年,單株抗體和重組蛋白生產應用將佔據最大的市場佔有率。單株抗體(mAb)已成為腫瘤學、感染疾病和罕見疾病等領域廣泛應用的生物治療方法。監管途徑已建立,生產流程已標準化,生物相似藥的競爭正在推動市場成長。標準化的多步驟純化流程為樹脂和耗材創造了穩定的需求。目前,業界以成熟的三步驟層析法法進行mAb純化:蛋白A親和性和捕獲(從細胞培養基中分離目標抗體)、中間純化(去除聚集體和殘留雜質,通常透過離子交換)和最終純化(達到規格級純度並去除殘留配體)。這些因素共同促成了單株抗體和重組蛋白生產應用領域中佔據最大的市場佔有率。
按最終用戶分類,製備和工藝層析法市場可細分為製藥和生物製藥公司、食品和營養補充劑公司、研究和學術機構以及其他最終用戶。 2024年,製藥和生物製藥公司佔最大市場。由於需要大規模生產品質穩定、產量高且符合法規要求的商業化生物製藥產品,一體化製藥和生物製藥企業是製備和製程層析法的主要基本客群。這些企業經營著專門的下游設施,這些設施針對單株抗體、重組蛋白和疫苗的純化進行了最佳化,因此需要持續供應樹脂、色譜柱、系統和技術支援。大型製藥企業擁有內部製程開發和生產方面的專業知識,並在先進的層析法平台上進行了大量投資。這些營運的穩定性和規模,加上嚴格的品質和法規要求,使得該公司作為關鍵最終用戶群擁有巨大的需求。這些因素促成了該公司佔據最大的市場佔有率。
製備和工藝層析法市場由五大區域組成:北美、歐洲、亞太、拉丁美洲以及中東和非洲。截至2025年,北美在製備和製程層析法產品市場佔據顯著佔有率。其主導地位歸功於北美生產能力的擴張,該能力專注於商業規模生產和臨床試驗供應。投資趨勢表明,美國聯邦和州政府的支持正在推動國內生物製造,以應對疫情並減少對國際供應鏈的依賴。中小型生物技術公司與合約研發生產機構(CDMO)合作的拓展也促成了北美在2025年佔據最大的市場佔有率。
製備和製程層析法市場的主要企業包括:丹納赫公司(美國)、Repligen公司(美國)、默克集團(德國)、賽默飛世爾科技(美國)、安捷倫科技公司(美國)、Bio-Rad Laboratories公司(美國)、Avantor公司(美國)、賽多利斯股份公司(德國)、島津公司(日本)、藝康公司(美國)、東曹生物科技(日本)、三菱化學集團公司(日本)、Revvity公司(美國)、珀金埃爾默公司(美國)、Cecil Instruments Can(英國)、赫西斯科尼亞公司(英國)、美國共和國公司(瑞士共和國)(瑞士共和國)(瑞士共和國)(瑞士共和國)公司(英國)、美國共和國)(瑞士共和國)(瑞士共和國)公司(瑞士共和國)公司(瑞士共和國)公司(瑞士共和國)公司(瑞士共和國公司(英國)、美國共和國)。 Science(天津)儀器技術有限公司(中國)、Bio Works Technologies公司(瑞典)、Sykam GmbH公司(德國)、Sunresin New Materials公司(中國)、YMC公司(日本)和Geno Technology Inc.公司(美國)。
調查範圍
本報告按產品、應用、最終用戶和地區分析製備色譜和工藝層析法市場。報告檢驗了推動市場成長的因素、跨產業挑戰和機遇,以及競爭格局,包括市場領導和中小企業。此外,報告中還包含五個地區各細分市場的收入預測和微觀市場分析。
為什麼要購買這份報告?
本報告透過提供製備色譜和製程層析法市場及其細分市場的準確市場收入預測,幫助市場領導和新參與企業。它幫助相關人員了解競爭格局,更有效地進行市場定位,並制定合適的打入市場策略。此外,報告還深入分析了市場動態,包括關鍵促進因素、限制因素、挑戰和機會。
該報告提供了以下方面的深入數據:
- 市場滲透率:對製備和工藝層析法市場主要企業提供的產品系列進行詳細分析
- 產品開發/創新:深入分析製備色譜和工藝層析法市場主要企業提供的產品系列
- 市場發展:關於盈利成長潛力領域的深刻洞察數據
- 市場多元化:了解製備色譜和工藝層析法市場的最新趨勢和進展
- 競爭格局評估:對主要競爭對手的產品、成長策略、收入預測和市場類別進行全面評估
目錄
第1章 引言
第2章執行摘要
第3章重要考察
第4章 市場概覽
- 市場動態
- 促進要素
- 抑制因素
- 機會
- 任務
- 未滿足的需求
- 相互關聯的市場與跨產業機遇
- 一級/二級/三級公司的策略性舉措
- 製備和工藝層析法市場參與者所採用策略概述
第5章 產業趨勢
- 波特五力分析
- 總體經濟指標
- 供應鏈分析
- 價值鏈分析
- 生態系分析
- 定價分析
- 貿易分析
- 2026-2027 年主要會議和活動
- 影響客戶業務的趨勢/顛覆性因素
- 投資和資金籌措方案
- 案例研究分析
- 2025年美國關稅對製備色譜和工藝層析法市場的影響
第6章:科技、專利和數位化/人工智慧應用帶來的策略顛覆
- 關鍵新興技術
- 互補技術
- 鄰近技術
- 專利分析
- 人工智慧對製備色譜和過程層析法市場的影響
- 成功案例和實際應用
第7章永續性和監管環境
- 監理環境與永續性舉措
- 監管機構、政府機構和其他組織
- 監管趨勢
- 業界標準
- 對永續性的承諾
- 監理政策如何影響永續性舉措
第8章:顧客狀況與購買行為
- 決策流程
- 買方相關人員和採購評估標準
- 招募障礙和內部挑戰
- 來自各個終端使用者產業的未滿足需求
- 市場盈利
9. 依產品類型分類的製備層析與製程層析法市場
- 工藝層析法產品
- 製備層析法產品
第10章:製備色譜和工藝層析法市場(按應用領域分類)
- 單株抗體和重組蛋白
- 疫苗
- 胜肽和寡核苷酸
- 細胞和基因治療
- 其他
11. 按最終用戶分類的製備和工藝層析法市場
- 製藥和生物製藥公司
- 食品和營養補充品公司
- 研究和學術機構
- 其他
12. 按地區分類的製備色譜和工藝層析法市場
- 北美洲
- 北美宏觀經濟展望
- 美國
- 加拿大
- 歐洲
- 歐洲宏觀經濟展望
- 德國
- 英國
- 法國
- 義大利
- 西班牙
- 其他
- 亞太地區
- 亞太宏觀經濟展望
- 日本
- 中國
- 印度
- 澳洲
- 韓國
- 其他
- 拉丁美洲
- 拉丁美洲宏觀經濟展望
- 巴西
- 墨西哥
- 其他
- 中東和非洲
- 中東和非洲宏觀經濟展望
- 海灣合作理事會國家
- 其他
第13章 競爭格局
- 主要參與企業的策略/優勢
- 2020-2024年收入分析
- 2025年市佔率分析
- 估值和財務指標
- 品牌/產品對比
- 公司估值矩陣:主要參與企業,2025 年
- 公司估值矩陣:Start-Ups/中小企業,2025 年
- 競爭場景
第14章:公司簡介
- 主要參與企業
- DANAHER
- THERMO FISHER SCIENTIFIC INC.
- MERCK KGAA
- SARTORIUS AG
- WATERS CORPORATION
- AGILENT TECHNOLOGIES, INC.
- AVANTOR, INC.
- BIO-RAD LABORATORIES, INC.
- REPLIGEN CORPORATION
- SHIMADZU CORPORATION
- ECOLAB INC.
- TOSOH BIOSCIENCE
- 其他公司
- MITSUBISHI CHEMICAL CORPORATION
- REVVITY
- PERKINELMER
- CECIL INSTRUMENTS LIMITED
- WR GRACE & CO.-CONN
- ECOM SPOL. S RO
- BUCHI LABORTECHNIK AG
- GOOD SCIENCE(TIANJIN)INSTRUMENT TECHNOLOGIES CO., LTD.
- BIO WORKS TECHNOLOGIES
- SYKAM GMBH
- SUNRESIN NEW MATERIALS CO. LTD.
- YMC CO., LTD.
- GENO TECHNOLOGY INC.
第15章調查方法
第16章附錄
The global preparative and process chromatography market is projected to reach USD 21.50 billion by 2031 from USD 14.32 billion in 2026, growing at a CAGR of 8.2% during the forecast period.
| Scope of the Report | |
|---|---|
| Years Considered for the Study | 2024-2031 |
| Base Year | 2025 |
| Forecast Period | 2025-2031 |
| Units Considered | Value (USD billion) |
| Segments | Product Type, Application, End User, and Region |
| Regions covered | North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa |
Vaccines and recombinant proteins using new modalities such as CGT, oligos, and mRNA are expected to grow in volume as demand for more personalized treatments rises in some developed economies. With increased volume, this diversity of biologics needs to be purified. These shifts make chromatography a critical unit operation, especially for capture and purification, creating space for market growth. The global monoclonal antibody (mAb) pipeline is expanding, marked by high FDA approvals and new bispecific and antibody-drug conjugate launches. This has helped boost purification requirements among end users, which is expected to increase the market for preparative and process chromatography products.

"Based on product type, process chromatography products held the largest share of the market in 2025."
Based on product, the preparative and process chromatography market is segmented into preparative chromatography and process chromatography. Process chromatography accounted for the largest share of the market. Global pharmaceutical and biopharmaceutical end users-which include major pharma facilities and CDMOs-are focused on commercial downstream workflows designed for process chromatography rather than preparative stages. New plants or large-scale manufacturing facilities drive recurring, multi-year demand for process resins, columns, and services. These factors helped process-scale chromatography products capture the largest share of the global market.
"Based on applications, the monoclonal antibodies and recombinant proteins segment held the largest share in 2025."
By application, the market for preparative and process chromatography is categorized into monoclonal antibodies & recombinant proteins, vaccines, peptides and oligonucleotides, cell and gene therapies, and other applications. The monoclonal antibodies & recombinant proteins production application acquired the largest market share in 2025. Monoclonal antibodies (mAbs) have become a widely adopted biotherapeutic modality across oncology, immunology, infectious disease, and rare disease indications. Regulatory pathways are well established, manufacturing processes are standardized, and biosimilar competition has driven market growth. Standardized, multi-step purification workflows drive consistent resin and consumables demand. The industry has converged on proven three-step chromatography trains for mAb purification: Protein A affinity capture (to isolate the target antibody from cell culture fluid), intermediate polishing (typically ion-exchange to remove aggregates and remaining impurities), and final polishing (to achieve specification-grade purity and remove residual ligands). These factors have helped the monoclonal antibodies & recombinant proteins production application segment acquire the largest share.
"Based on end user, the pharma and biopharma companies segment accounted for the largest share in 2025."
By end user, the preparative and process chromatography market is categorized into pharma & biopharma companies, food & nutraceutical companies, research & academia, and other end users. Pharma & biopharma companies held the largest share in 2024. Integrated pharmaceutical and biopharmaceutical manufacturers represent the anchor customer base for preparative and process chromatography, driven by their need to produce large-scale commercial biologics with consistent quality, high yield, and regulatory compliance. These organizations operate dedicated downstream facilities optimized for mAb, recombinant protein, and vaccine purification, requiring a continuous supply of resins, columns, systems, and technical support. Large pharma companies maintain in-house process development and manufacturing expertise and invest heavily in advanced chromatography platforms. The stability and scale of these operations, combined with their strict quality and regulatory requirements, make them a key end user segment with significant demand. These factors have helped them acquire the largest share.
"North America is expected to hold a significant share in the preparative and process chromatography market throughout the forecast period."
The preparative and process chromatography market comprises five main regions: North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa. In 2025, North America held a significant share of the market for preparative and process chromatography products. Its dominant position stems from increased manufacturing capacity focused on both commercial-scale and clinical-trial supply production. Investment trends indicate that US federal and state support promotes domestic biomanufacturing for pandemic preparedness and to reduce reliance on international supply chains. Small- to mid-sized biotech firms are increasingly partnering with CDMOs. These factors helped North America secure the largest market share in 2025.
A breakdown of the primary participants referred to for this report is provided below:
- By Company Type: Tier 1 (35%), Tier 2 (45%), and Tier 3 (20%)
- By Designation: C-level Executives (35%), Directors (25%), and Others (40%)
- By Region: North America (40%), Europe (30%), Asia Pacific (20%), Latin America (5%), and the Middle East & Africa (5%)
The key players profiled in the preparative and process chromatography market are Danaher Corporation (US), Repligen Corporation (US), Merck KGaA (Germany), Thermo Fisher Scientific (US), Agilent Technologies, Inc. (US), Bio-Rad Laboratories, Inc. (US), Avantor, Inc. (US), Sartorius AG (Germany), Shimadzu Corporation (Japan), Ecolab Inc. (US), Tosoh Bioscience (Japan), Mitsubishi Chemical Group Corporation (Japan), Revvity (US), PerkinElmer (US), Cecil Instruments Limited (UK), W.R. Grace & Co. (US), Ecom SPO (Czech Republic), BUCHI Labortechnik AG (Switzerland), Good Science (Tianjin) Instrument Technologies Co., Ltd. (China), Bio Works Technologies (Sweden), Sykam GmbH (Germany), Sunresin New Materials Co., Ltd. (China), YMC Co., Ltd. (Japan), and Geno Technology Inc. (US)
Research Coverage
The research report analyzes the preparative and process chromatography market by product, application, end user, and region. It examines the factors driving market growth, the challenges and opportunities across industries, and the competitive landscape, including market leaders and small- to medium-sized enterprises. It also estimates revenue for different market segments across five regions and includes a micromarket analysis.
Reasons to Buy the Report
The report will help market leaders and new entrants by providing accurate revenue estimates for the entire preparative and process chromatography market and its subsegments. It will help stakeholders understand the competitive landscape, enabling them to position their businesses more effectively and develop suitable go-to-market strategies. Additionally, the report offers insights into market dynamics, including key drivers, restraints, challenges, and opportunities.
This report provides insightful data on the following.
- Market Penetration: In-depth coverage of product portfolios offered by the top players in the preparative and process chromatography market
- Product Development/Innovation: In-depth coverage of product portfolios offered by the top players in the preparative and process chromatography market
- Market Development: Insightful data on profitable developing areas
- Market Diversification: Details about recent developments and advancements in the preparative and process chromatography market
- Competitive Assessment: Extensive assessment of the products, growth tactics, revenue projections, and market categories of the top competitors
TABLE OF CONTENTS
1 INTRODUCTION
- 1.1 STUDY OBJECTIVES
- 1.2 MARKET DEFINITION
- 1.3 STUDY SCOPE
- 1.3.1 MARKET SEGMENTATION & REGIONAL SCOPE
- 1.3.2 INCLUSIONS & EXCLUSIONS
- 1.3.3 YEARS CONSIDERED
- 1.4 CURRENCY CONSIDERED
- 1.5 STAKEHOLDERS
- 1.6 SUMMARY OF CHANGES
2 EXECUTIVE SUMMARY
- 2.1 MARKET HIGHLIGHTS & KEY INSIGHTS
- 2.2 KEY MARKET PARTICIPANTS: MAPPING OF STRATEGIC DEVELOPMENTS
- 2.3 DISRUPTIVE TRENDS IN PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET
- 2.4 HIGH-GROWTH SEGMENTS
- 2.5 REGIONAL SNAPSHOT: MARKET SIZE, GROWTH RATE, AND FORECAST
3 PREMIUM INSIGHTS
- 3.1 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET OVERVIEW
- 3.2 ASIA PACIFIC PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, BY END USER AND COUNTRY
- 3.3 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: GEOGRAPHIC GROWTH OPPORTUNITIES
4 MARKET OVERVIEW
- 4.1 INTRODUCTION
- 4.2 MARKET DYNAMICS
- 4.2.1 DRIVERS
- 4.2.1.1 Increased demand for complex biologics and advanced therapeutic modalities
- 4.2.1.2 Increased outsourcing to CDMOs
- 4.2.1.3 Technological advancements in chromatography media and systems
- 4.2.1.4 Need for cost optimization in downstream processing
- 4.2.2 RESTRAINTS
- 4.2.2.1 Lowered replacement rate due to incomplete single-use adoption
- 4.2.2.2 Limited resin lifetime and performance degradation
- 4.2.3 OPPORTUNITIES
- 4.2.3.1 Ongoing investments in bioprocessing capacity and analytical capabilities
- 4.2.3.2 Expansion of integrated, intensified, and continuous chromatography columns
- 4.2.4 CHALLENGES
- 4.2.4.1 Shortage of skilled chromatography professionals
- 4.2.4.2 High capital and operating costs
- 4.2.1 DRIVERS
- 4.3 UNMET NEEDS
- 4.4 INTERCONNECTED MARKETS & CROSS-SECTOR OPPORTUNITIES
- 4.5 STRATEGIC MOVES BY TIER1/2/3 PLAYERS
- 4.5.1 OVERVIEW OF STRATEGIES ADOPTED BY PLAYERS IN PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET
5 INDUSTRY TRENDS
- 5.1 PORTER'S FIVE FORCES ANALYSIS
- 5.1.1 BARGAINING POWER OF BUYERS
- 5.1.2 BARGAINING POWER OF SUPPLIERS
- 5.1.3 THREAT OF NEW ENTRANTS
- 5.1.4 THREAT OF SUBSTITUTES
- 5.1.5 INTENSITY OF COMPETITIVE RIVALRY
- 5.2 MACROECONOMIC INDICATORS
- 5.2.1 INTRODUCTION
- 5.2.2 GDP TRENDS & FORECAST
- 5.2.3 TRENDS IN GLOBAL BIOPROCESS INDUSTRY
- 5.3 SUPPLY CHAIN ANALYSIS
- 5.3.1 PROMINENT COMPANIES
- 5.3.2 SMALL & MEDIUM-SIZED ENTERPRISES
- 5.3.3 END USERS
- 5.4 VALUE CHAIN ANALYSIS
- 5.4.1 RESEARCH & PRODUCT DEVELOPMENT
- 5.4.2 RAW MATERIAL PROCUREMENT
- 5.4.3 MANUFACTURING
- 5.4.4 DISTRIBUTION, MARKETING & SALES, AND POST-SALES SERVICES
- 5.5 ECOSYSTEM ANALYSIS
- 5.6 PRICING ANALYSIS
- 5.6.1 AVERAGE SELLING PRICE OF PREPARATIVE AND PROCESS CHROMATOGRAPHY PRODUCTS, BY KEY PLAYER, 2025
- 5.6.2 AVERAGE SELLING PRICE OF PREPARATIVE AND PROCESS CHROMATOGRAPHY PRODUCTS, BY REGION, 2025
- 5.7 TRADE ANALYSIS
- 5.7.1 IMPORT DATA (HS CODE 902720)
- 5.7.2 EXPORT DATA (HS CODE 902720)
- 5.8 KEY CONFERENCES & EVENTS, 2026-2027
- 5.9 TRENDS/DISRUPTIONS IMPACTING CUSTOMERS' BUSINESSES
- 5.10 INVESTMENT & FUNDING SCENARIO
- 5.11 CASE STUDY ANALYSIS
- 5.12 IMPACT OF 2025 US TARIFFS ON PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET
- 5.12.1 INTRODUCTION
- 5.12.2 KEY TARIFF RATES
- 5.12.3 PRICE IMPACT ANALYSIS
- 5.12.4 IMPACT ON COUNTRY/REGION
- 5.12.4.1 US
- 5.12.4.2 Europe
- 5.12.4.3 Asia Pacific
- 5.12.5 IMPACT ON END-USE INDUSTRIES
6 STRATEGIC DISRUPTIONS THROUGH TECHNOLOGY, PATENTS, AND DIGITAL & AI ADOPTION
- 6.1 KEY EMERGING TECHNOLOGIES
- 6.1.1 RECOMBINANT PROTEIN A LIGAND ENGINEERING
- 6.1.2 ALKALINE-STABLE LIGAND DEVELOPMENT
- 6.2 COMPLEMENTARY TECHNOLOGIES
- 6.2.1 AUTOMATED COLUMN PACKING SYSTEMS
- 6.2.2 IN-LINE MONITORING AND PAT (PROCESS ANALYTICAL TECHNOLOGY)
- 6.3 ADJACENT TECHNOLOGIES
- 6.3.1 CONTINUOUS CHROMATOGRAPHY SYSTEMS
- 6.3.2 AFFINITY MEMBRANE TECHNOLOGY
- 6.4 PATENT ANALYSIS
- 6.5 IMPACT OF AI ON PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET
- 6.5.1 TOP USE CASES & MARKET POTENTIAL
- 6.5.2 BEST PRACTICES IN PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET
- 6.5.3 CLIENTS' READINESS TO ADOPT GENERATIVE AI IN PREPARATIVE AND PROCESS CHROMATOGRAPHY
- 6.6 SUCCESS STORIES & REAL-WORLD APPLICATIONS
7 SUSTAINABILITY & REGULATORY LANDSCAPE
- 7.1 REGULATORY LANDSCAPE & SUSTAINABILITY INITIATIVES
- 7.1.1 REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- 7.1.2 REGULATORY TRENDS
- 7.1.2.1 North America
- 7.1.2.1.1 US
- 7.1.2.2 Europe
- 7.1.2.3 Asia Pacific
- 7.1.2.4 Rest of the World
- 7.1.2.1 North America
- 7.2 INDUSTRY STANDARDS
- 7.2.1 SUSTAINABILITY INITIATIVES
- 7.2.2 IMPACT OF REGULATORY POLICIES ON SUSTAINABILITY INITIATIVES
8 CUSTOMER LANDSCAPE & BUYER BEHAVIOR
- 8.1 DECISION-MAKING PROCESS
- 8.2 BUYER STAKEHOLDERS & BUYING EVALUATION CRITERIA
- 8.2.1 KEY STAKEHOLDERS IN BUYING PROCESS
- 8.2.2 BUYING EVALUATION CRITERIA
- 8.3 ADOPTION BARRIERS & INTERNAL CHALLENGES
- 8.4 UNMET NEEDS FROM VARIOUS END-USE INDUSTRIES
- 8.5 MARKET PROFITABILITY
9 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, BY PRODUCT TYPE
- 9.1 INTRODUCTION
- 9.2 PROCESS CHROMATOGRAPHY PRODUCTS
- 9.2.1 CHEMICALS & REAGENTS
- 9.2.1.1 Recurring consumption to drive demand for chemicals & reagents
- 9.2.2 RESINS
- 9.2.2.1 Increased purification volume to drive demand for resins
- 9.2.3 COLUMNS
- 9.2.3.1 Reproducible results and low cross-contamination risk to drive demand for columns in process chromatography operations
- 9.2.4 SYSTEMS
- 9.2.4.1 Less changeover time and maintenance needs to boost demand for single-use chromatography systems
- 9.2.5 AFTER-MARKET SERVICES
- 9.2.5.1 Capacity expansions and routine calibration needs to drive market growth
- 9.2.1 CHEMICALS & REAGENTS
- 9.3 PREPARATIVE CHROMATOGRAPHY PRODUCTS
- 9.3.1 CHEMICALS & REAGENTS
- 9.3.1.1 Method development focus and high-purity results to drive market for reagents in preparative operations
- 9.3.2 RESINS
- 9.3.2.1 Cost-effective supply chain metrics to drive demand for bulk resins
- 9.3.3 COLUMNS
- 9.3.3.1 Flexibility, low sample consumption, and high throughput to drive adoption
- 9.3.4 SYSTEMS
- 9.3.4.1 Biologics innovation and strong mAb pipeline to support demand for systems
- 9.3.5 AFTER-MARKET SERVICES
- 9.3.5.1 Consultative and technical services to drive demand for after-market services
- 9.3.1 CHEMICALS & REAGENTS
10 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, BY APPLICATION
- 10.1 INTRODUCTION
- 10.2 MONOCLONAL ANTIBODIES & RECOMBINANT PROTEINS
- 10.2.1 SHIFT TOWARDS MORE COST-EFFECTIVE OPTIONS TO OPEN NEW REVENUE CHANNELS
- 10.3 VACCINES
- 10.3.1 WIDE APPLICATION BASE OF STANDARDIZED CHROMATOGRAPHY TECHNIQUES IN VACCINE MANUFACTURING TO DRIVE MARKET GROWTH
- 10.4 PEPTIDES & OLIGONUCLEOTIDES
- 10.4.1 SHIFT TOWARDS HYBRID SYNTHESIS TO DRIVE MARKET GROWTH
- 10.5 CELL & GENE THERAPIES
- 10.5.1 LOWER TARGET PRODUCT YIELD TO HAMPER GROWTH OF CHROMATOGRAPHY AS PRIMARY TECHNIQUE
- 10.6 OTHER APPLICATIONS
11 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, BY END USER
- 11.1 INTRODUCTION
- 11.2 PHARMACEUTICAL & BIOPHARMACEUTICAL COMPANIES
- 11.2.1 EXTENSIVE USE IN BIOLOGICAL DRUG PRODUCTION TO ENSURE STRONG DEMAND
- 11.3 FOOD & NUTRACEUTICAL COMPANIES
- 11.3.1 EXPANDING USE CASES IN NUTRACEUTICAL INDUSTRY TO BOOST MARKET GROWTH
- 11.4 RESEARCH & ACADEMIC INSTITUTES
- 11.4.1 INCREASING R&D IN DRUG DISCOVERY TO DRIVE USE OF CHROMATOGRAPHY
- 11.5 OTHER END USERS
12 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, BY REGION
- 12.1 INTRODUCTION
- 12.2 NORTH AMERICA
- 12.2.1 MACROECONOMIC OUTLOOK FOR NORTH AMERICA
- 12.2.2 US
- 12.2.2.1 Expansion in biomanufacturing and domestic capacity to drive market
- 12.2.3 CANADA
- 12.2.3.1 Policy-led capacity build and CDMO growth to drive market
- 12.3 EUROPE
- 12.3.1 MACROECONOMIC OUTLOOK FOR EUROPE
- 12.3.2 GERMANY
- 12.3.2.1 Capacity expansion in end-use facilities to generate new demand channels for target products
- 12.3.3 UK
- 12.3.3.1 Government policies and public-private partnerships to drive market
- 12.3.4 FRANCE
- 12.3.4.1 Demand-side developments to add new revenue channels for chromatography OEMs and suppliers
- 12.3.5 ITALY
- 12.3.5.1 Increased CAPEX from major end-user categories to fuel demand for target products
- 12.3.6 SPAIN
- 12.3.6.1 Increased number of CDMOs and CROs to propel demand for chromatography and related products
- 12.3.7 REST OF EUROPE
- 12.4 ASIA PACIFIC
- 12.4.1 MACROECONOMIC OUTLOOK FOR ASIA PACIFIC
- 12.4.2 JAPAN
- 12.4.2.1 CDMO capacity expansions, policy backing, and CDMO investments to propel market
- 12.4.3 CHINA
- 12.4.3.1 Robust biopharmaceutical expansion to support demand
- 12.4.4 INDIA
- 12.4.4.1 Growth of pharma and biotech industries to drive demand for preparative and process chromatography
- 12.4.5 AUSTRALIA
- 12.4.5.1 Increasing demand for protein-based therapeutics to drive growth
- 12.4.6 SOUTH KOREA
- 12.4.6.1 Developments in biopharmaceutical sector to drive market growth
- 12.4.7 REST OF ASIA PACIFIC
- 12.5 LATIN AMERICA
- 12.5.1 MACROECONOMIC OUTLOOK FOR LATIN AMERICA
- 12.5.2 BRAZIL
- 12.5.2.1 Biomanufacturing emphasis and public-private partnerships to catalyze demand
- 12.5.3 MEXICO
- 12.5.3.1 Supportive government initiatives to boost demand for preparative and process chromatography products
- 12.5.4 REST OF LATIN AMERICA
- 12.6 MIDDLE EAST & AFRICA
- 12.6.1 MACROECONOMIC OUTLOOK FOR MIDDLE EAST & AFRICA
- 12.6.2 GCC COUNTRIES
- 12.6.2.1 Favorable government policies to propel market growth
- 12.6.3 REST OF MIDDLE EAST & AFRICA
13 COMPETITIVE LANDSCAPE
- 13.1 INTRODUCTION
- 13.2 KEY PLAYER STRATEGIES/RIGHT TO WIN
- 13.2.1 OVERVIEW OF STRATEGIES ADOPTED BY KEY PLAYERS IN PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET
- 13.3 REVENUE ANALYSIS, 2020-2024
- 13.4 MARKET SHARE ANALYSIS, 2025
- 13.4.1 MARKET RANKING OF KEY PLAYERS, 2025
- 13.5 COMPANY VALUATION & FINANCIAL METRICS
- 13.5.1 FINANCIAL METRICS
- 13.5.2 COMPANY VALUATION
- 13.6 BRAND/PRODUCT COMPARISON
- 13.7 COMPANY EVALUATION MATRIX: KEY PLAYERS, 2025
- 13.7.1 STARS
- 13.7.2 EMERGING LEADERS
- 13.7.3 PERVASIVE PLAYERS
- 13.7.4 PARTICIPANTS
- 13.7.5 COMPANY FOOTPRINT: KEY PLAYERS, 2025
- 13.7.5.1 Company footprint
- 13.7.5.2 Region footprint
- 13.7.5.3 Product type footprint
- 13.7.5.4 Application footprint
- 13.7.5.5 End-user footprint
- 13.8 COMPANY EVALUATION MATRIX: STARTUPS/SMES, 2025
- 13.8.1 PROGRESSIVE COMPANIES
- 13.8.2 RESPONSIVE COMPANIES
- 13.8.3 DYNAMIC COMPANIES
- 13.8.4 STARTING BLOCKS
- 13.8.5 COMPETITIVE BENCHMARKING OF STARTUPS/SMES, 2025
- 13.8.5.1 Detailed list of key startups/SMEs
- 13.8.5.2 Competitive benchmarking of key startups/SMEs
- 13.9 COMPETITIVE SCENARIO
- 13.9.1 PRODUCT LAUNCHES
- 13.9.2 DEALS
- 13.9.3 EXPANSIONS
14 COMPANY PROFILES
- 14.1 KEY PLAYERS
- 14.1.1 DANAHER
- 14.1.1.1 Business overview
- 14.1.1.2 Products offered
- 14.1.1.3 Recent developments
- 14.1.1.3.1 Product launches
- 14.1.1.3.2 Deals
- 14.1.1.3.3 Expansions
- 14.1.1.4 MnM view
- 14.1.1.4.1 Right to win
- 14.1.1.4.2 Strategic choices
- 14.1.1.4.3 Weaknesses & competitive threats
- 14.1.2 THERMO FISHER SCIENTIFIC INC.
- 14.1.2.1 Business overview
- 14.1.2.2 Products offered
- 14.1.2.3 Recent developments
- 14.1.2.3.1 Deals
- 14.1.2.3.2 Expansions
- 14.1.2.4 MnM view
- 14.1.2.4.1 Right to win
- 14.1.2.4.2 Strategic choices
- 14.1.2.4.3 Weaknesses & competitive threats
- 14.1.3 MERCK KGAA
- 14.1.3.1 Business overview
- 14.1.3.2 Products offered
- 14.1.3.3 Recent developments
- 14.1.3.3.1 Deals
- 14.1.3.3.2 Expansions
- 14.1.3.3.3 Other developments
- 14.1.3.4 MnM view
- 14.1.3.4.1 Right to win
- 14.1.3.4.2 Strategic choices
- 14.1.3.4.3 Weaknesses & competitive threats
- 14.1.4 SARTORIUS AG
- 14.1.4.1 Business overview
- 14.1.4.2 Products offered
- 14.1.4.3 MnM view
- 14.1.4.3.1 Right to win
- 14.1.4.3.2 Strategic choices
- 14.1.4.3.3 Weaknesses & competitive threats
- 14.1.5 WATERS CORPORATION
- 14.1.5.1 Business overview
- 14.1.5.2 Products offered
- 14.1.5.3 Recent developments
- 14.1.5.3.1 Product launches
- 14.1.5.3.2 Deals
- 14.1.5.4 MnM view
- 14.1.5.4.1 Right to win
- 14.1.5.4.2 Strategic choices
- 14.1.5.4.3 Weaknesses & competitive threats
- 14.1.6 AGILENT TECHNOLOGIES, INC.
- 14.1.6.1 Business overview
- 14.1.6.2 Products offered
- 14.1.6.3 Recent developments
- 14.1.6.3.1 Product launches
- 14.1.6.3.2 Deals
- 14.1.6.3.3 Expansions
- 14.1.7 AVANTOR, INC.
- 14.1.7.1 Business overview
- 14.1.7.2 Products offered
- 14.1.8 BIO-RAD LABORATORIES, INC.
- 14.1.8.1 Business overview
- 14.1.8.2 Products offered
- 14.1.9 REPLIGEN CORPORATION
- 14.1.9.1 Business overview
- 14.1.9.2 Products offered
- 14.1.9.3 Recent developments
- 14.1.9.3.1 Product launches
- 14.1.9.3.2 Deals
- 14.1.10 SHIMADZU CORPORATION
- 14.1.10.1 Business overview
- 14.1.10.2 Products offered
- 14.1.10.3 Recent developments
- 14.1.10.3.1 Deals
- 14.1.11 ECOLAB INC.
- 14.1.11.1 Business overview
- 14.1.11.2 Products offered
- 14.1.11.3 Recent developments
- 14.1.11.3.1 Product launches
- 14.1.11.3.2 Deals
- 14.1.11.3.3 Expansions
- 14.1.12 TOSOH BIOSCIENCE
- 14.1.12.1 Business overview
- 14.1.12.2 Products offered
- 14.1.12.3 Recent developments
- 14.1.12.3.1 Expansions
- 14.1.1 DANAHER
- 14.2 OTHER PLAYERS
- 14.2.1 MITSUBISHI CHEMICAL CORPORATION
- 14.2.2 REVVITY
- 14.2.3 PERKINELMER
- 14.2.4 CECIL INSTRUMENTS LIMITED
- 14.2.5 W. R. GRACE & CO.-CONN
- 14.2.6 ECOM SPOL. S R.O
- 14.2.7 BUCHI LABORTECHNIK AG
- 14.2.8 GOOD SCIENCE (TIANJIN) INSTRUMENT TECHNOLOGIES CO., LTD.
- 14.2.9 BIO WORKS TECHNOLOGIES
- 14.2.10 SYKAM GMBH
- 14.2.11 SUNRESIN NEW MATERIALS CO. LTD.
- 14.2.12 YMC CO., LTD.
- 14.2.13 GENO TECHNOLOGY INC.
15 RESEARCH METHODOLOGY
- 15.1 RESEARCH DATA
- 15.1.1 SECONDARY RESEARCH
- 15.1.1.1 Key secondary sources
- 15.1.1.2 Key data from secondary sources
- 15.1.1.3 Objectives of secondary research
- 15.1.2 PRIMARY RESEARCH
- 15.1.2.1 Key primary sources
- 15.1.2.2 Key supply- and demand-side participants
- 15.1.2.3 Breakdown of primary interviews
- 15.1.2.4 Objectives of primary research
- 15.1.2.5 Key primary insights
- 15.1.1 SECONDARY RESEARCH
- 15.2 MARKET SIZE ESTIMATION
- 15.2.1 BOTTOM-UP APPROACH
- 15.2.1.1 Company revenue estimation
- 15.2.1.2 Customer-based market estimation
- 15.2.1.3 Primary interviews
- 15.2.2 TOP-DOWN APPROACH
- 15.2.1 BOTTOM-UP APPROACH
- 15.3 GROWTH RATE ASSUMPTIONS
- 15.4 DATA TRIANGULATION
- 15.5 STUDY ASSUMPTIONS
- 15.6 RESEARCH LIMITATIONS
- 15.7 RISK ANALYSIS
16 APPENDIX
- 16.1 DISCUSSION GUIDE
- 16.2 KNOWLEDGESTORE: MARKETSANDMARKETS' SUBSCRIPTION PORTAL
- 16.3 CUSTOMIZATION OPTIONS
- 16.4 RELATED REPORTS
- 16.5 AUTHOR DETAILS
List of Tables
- TABLE 1 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: INCLUSIONS & EXCLUSIONS
- TABLE 2 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: UNMET NEEDS
- TABLE 3 OVERVIEW OF STRATEGIES ADOPTED BY KEY COMPANIES IN PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET
- TABLE 4 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: PORTER'S FIVE FORCES ANALYSIS
- TABLE 5 AVERAGE SELLING PRICE OF GC COLUMNS, BY KEY PLAYER, 2025 (USD)
- TABLE 6 AVERAGE SELLING PRICE TREND OF PREPARATIVE AND PROCESS CHROMATOGRAPHY PRODUCTS, BY REGION, 2023-2025 (USD)
- TABLE 7 IMPORT DATA FOR CHROMATOGRAPHS AND ELECTROPHORESIS INSTRUMENTS, BY COUNTRY, 2021-2024 (USD THOUSAND)
- TABLE 8 EXPORT DATA FOR CHROMATOGRAPHS AND ELECTROPHORESIS INSTRUMENTS, BY COUNTRY, 2021-2024 (USD THOUSAND)
- TABLE 9 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: LIST OF MAJOR CONFERENCES AND EVENTS
- TABLE 10 CASE STUDY 1: BIO-WORKS TECHNOLOGIES - WORKBEADS AFFIMAB FOR MONOCLONAL ANTIBODY PURIFICATION
- TABLE 11 CASE STUDY 2: STATISTICAL APPROACHES TO DEVELOPMENT OF PROTEIN A AFFINITY CHROMATOGRAPHY FOR IMPROVING RECOVERY AND CONTROLLING QUALITY ATTRIBUTES OF MONOCLONAL ANTIBODIES
- TABLE 12 CASE STUDY 3: CAPTURE CHROMATOGRAPHY WITH MIXED-MODE RESINS: A CASE STUDY WITH RECOMBINANT HUMAN THIOREDOXIN FROM ESCHERICHIA COLI
- TABLE 13 US-ADJUSTED RECIPROCAL TARIFF RATES
- TABLE 14 CASE STUDY 1: AI-GENERATED VS. IN-LAB HPLC METHOD OPTIMIZATION
- TABLE 15 INTERCONNECTED ADJACENT ECOSYSTEMS & IMPACT ON MARKET PLAYERS
- TABLE 16 NORTH AMERICA: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 17 EUROPE: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 18 ASIA PACIFIC: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 19 LATIN AMERICA: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 20 MIDDLE EAST & AFRICA: REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- TABLE 21 INDUSTRY STANDARDS FOR PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET
- TABLE 22 INFLUENCE OF STAKEHOLDERS ON BUYING PROCESS (%)
- TABLE 23 KEY BUYING CRITERIA, BY END USER
- TABLE 24 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 25 PROCESS CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 26 PROCESS CHROMATOGRAPHY PRODUCTS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 27 PROCESS CHROMATOGRAPHY PRODUCTS MARKET FOR CHEMICALS & REAGENTS, BY REGION, 2024-2031 (USD MILLION)
- TABLE 28 PROCESS CHROMATOGRAPHY PRODUCTS MARKET FOR RESINS, BY REGION, 2024-2031 (USD MILLION)
- TABLE 29 PROCESS CHROMATOGRAPHY PRODUCTS MARKET FOR COLUMNS, BY REGION, 2024-2031 (USD MILLION)
- TABLE 30 PROCESS CHROMATOGRAPHY PRODUCTS MARKET FOR SYSTEMS, BY REGION, 2024-2031 (USD MILLION)
- TABLE 31 PROCESS CHROMATOGRAPHY PRODUCTS MARKET FOR AFTER-MARKET SERVICES, BY REGION, 2024-2031 (USD MILLION)
- TABLE 32 PREPARATIVE CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 33 PREPARATIVE CHROMATOGRAPHY PRODUCTS MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 34 PREPARATIVE CHROMATOGRAPHY PRODUCTS MARKET FOR CHEMICALS & REAGENTS, BY REGION, 2024-2031 (USD MILLION)
- TABLE 35 PREPARATIVE CHROMATOGRAPHY PRODUCTS MARKET FOR RESINS, BY REGION, 2024-2031 (USD MILLION)
- TABLE 36 PREPARATIVE CHROMATOGRAPHY PRODUCTS MARKET FOR COLUMNS, BY REGION, 2024-2031 (USD MILLION)
- TABLE 37 PREPARATIVE CHROMATOGRAPHY PRODUCTS MARKET FOR SYSTEMS, BY REGION, 2024-2031 (USD MILLION)
- TABLE 38 PREPARATIVE CHROMATOGRAPHY PRODUCTS MARKET FOR AFTER-MARKET SERVICES, BY REGION, 2024-2031 (USD MILLION)
- TABLE 39 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 40 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET FOR MONOCLONAL ANTIBODIES & RECOMBINANT PROTEINS, BY REGION, 2024-2031 (USD MILLION)
- TABLE 41 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET FOR VACCINES, BY REGION, 2024-2031 (USD MILLION)
- TABLE 42 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET FOR PEPTIDES & OLIGONUCLEOTIDES, BY REGION, 2024-2031 (USD MILLION)
- TABLE 43 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET FOR CELL & GENE THERAPIES, BY REGION, 2024-2031 (USD MILLION)
- TABLE 44 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET FOR OTHER APPLICATIONS, BY REGION, 2024-2031 (USD MILLION)
- TABLE 45 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 46 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET FOR PHARMACEUTICAL & BIOPHARMACEUTICAL COMPANIES, BY REGION, 2024-2031 (USD MILLION)
- TABLE 47 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET FOR FOOD & NUTRACEUTICAL COMPANIES, BY REGION, 2024-2031 (USD MILLION)
- TABLE 48 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET FOR RESEARCH & ACADEMIC INSTITUTES, BY REGION, 2024-2031 (USD MILLION)
- TABLE 49 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET FOR OTHER END USERS, BY REGION, 2024-2031 (USD MILLION)
- TABLE 50 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 51 NORTH AMERICA: KEY MACROINDICATORS
- TABLE 52 NORTH AMERICA: PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 53 NORTH AMERICA: PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 54 NORTH AMERICA: PROCESS CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 55 NORTH AMERICA: PREPARATIVE CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 56 NORTH AMERICA: PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 57 NORTH AMERICA: PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 58 US: PROCESS CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 59 US: PREPARATIVE CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 60 CANADA: PROCESS CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 61 CANADA: PREPARATIVE CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 62 EUROPE: KEY MACROINDICATORS
- TABLE 63 EUROPE: PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 64 EUROPE: PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 65 EUROPE: PROCESS CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 66 EUROPE: PREPARATIVE CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 67 EUROPE: PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 68 EUROPE: PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 69 GERMANY: PROCESS CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 70 GERMANY: PREPARATIVE CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 71 UK: PROCESS CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 72 UK: PREPARATIVE CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 73 FRANCE: PROCESS CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 74 FRANCE: PREPARATIVE CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 75 ITALY: PROCESS CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 76 ITALY: PREPARATIVE CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 77 SPAIN: PROCESS CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 78 SPAIN: PREPARATIVE CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 79 REST OF EUROPE: PROCESS CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 80 REST OF EUROPE: PREPARATIVE CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 81 ASIA PACIFIC: KEY MACROINDICATORS
- TABLE 82 ASIA PACIFIC: PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 83 ASIA PACIFIC: PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 84 ASIA PACIFIC: PROCESS CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 85 ASIA PACIFIC: PREPARATIVE CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 86 ASIA PACIFIC: PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 87 ASIA PACIFIC: PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 88 JAPAN: PROCESS CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 89 JAPAN: PREPARATIVE CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 90 CHINA: PROCESS CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 91 CHINA: PREPARATIVE CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 92 INDIA: PROCESS CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 93 INDIA: PREPARATIVE CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 94 AUSTRALIA: PROCESS CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 95 AUSTRALIA: PREPARATIVE CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 96 SOUTH KOREA: PROCESS CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 97 SOUTH KOREA: PREPARATIVE CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 98 REST OF ASIA PACIFIC: PROCESS CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 99 REST OF ASIA PACIFIC: PREPARATIVE CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 100 LATIN AMERICA: KEY MACROINDICATORS
- TABLE 101 LATIN AMERICA: PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, BY COUNTRY, 2024-2031 (USD MILLION)
- TABLE 102 LATIN AMERICA: PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 103 LATIN AMERICA: PROCESS CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 104 LATIN AMERICA: PREPARATIVE CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 105 LATIN AMERICA: PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 106 LATIN AMERICA: PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 107 BRAZIL: PROCESS CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 108 BRAZIL: PREPARATIVE CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 109 MEXICO: PROCESS CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 110 MEXICO: PREPARATIVE CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 111 REST OF LATIN AMERICA: PROCESS CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 112 REST OF LATIN AMERICA: PREPARATIVE CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 113 MIDDLE EAST & AFRICA: KEY MACROINDICATORS
- TABLE 114 MIDDLE EAST & AFRICA: PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, BY REGION, 2024-2031 (USD MILLION)
- TABLE 115 MIDDLE EAST & AFRICA: PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, BY PRODUCT TYPE, 2024-2031 (USD MILLION)
- TABLE 116 MIDDLE EAST & AFRICA: PROCESS CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 117 MIDDLE EAST & AFRICA: PREPARATIVE CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 118 MIDDLE EAST & AFRICA: PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, BY APPLICATION, 2024-2031 (USD MILLION)
- TABLE 119 MIDDLE EAST & AFRICA: PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, BY END USER, 2024-2031 (USD MILLION)
- TABLE 120 GCC COUNTRIES: PROCESS CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 121 GCC COUNTRIES: PREPARATIVE CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 122 REST OF MIDDLE EAST & AFRICA: PROCESS CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 123 REST OF MIDDLE EAST & AFRICA: PREPARATIVE CHROMATOGRAPHY PRODUCTS MARKET, BY TYPE, 2024-2031 (USD MILLION)
- TABLE 124 OVERVIEW OF STRATEGIES DEPLOYED BY KEY PLAYERS IN PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, 2022-2025
- TABLE 125 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: DEGREE OF COMPETITION
- TABLE 126 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: REGION FOOTPRINT
- TABLE 127 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: PRODUCT TYPE FOOTPRINT
- TABLE 128 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: APPLICATION FOOTPRINT
- TABLE 129 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: END-USER FOOTPRINT
- TABLE 130 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: DETAILED LIST OF KEY STARTUPS/SME PLAYERS, 2025
- TABLE 131 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: COMPETITIVE BENCHMARKING OF KEY STARTUPS/SME PLAYERS, BY PRODUCT TYPE AND REGION
- TABLE 132 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: PRODUCT LAUNCHES, JANUARY 2022-JANUARY 2026
- TABLE 133 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: DEALS, JANUARY 2022-JANUARY 2026
- TABLE 134 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: EXPANSIONS, JANUARY 2022-JANUARY 2026
- TABLE 135 DANAHER: COMPANY OVERVIEW
- TABLE 136 DANAHER: PRODUCTS OFFERED
- TABLE 137 DANAHER: PRODUCT LAUNCHES, JANUARY 2022-DECEMBER 2025
- TABLE 138 DANAHER: DEALS, JANUARY 2022-DECEMBER 2025
- TABLE 139 DANAHER: EXPANSIONS, JANUARY 2022-JULY 2025
- TABLE 140 THERMO FISHER SCIENTIFIC INC.: COMPANY OVERVIEW
- TABLE 141 THERMO FISHER SCIENTIFIC INC.: PRODUCTS OFFERED
- TABLE 142 THERMO FISHER SCIENTIFIC INC.: DEALS, JANUARY 2022-SEPTEMBER 2025
- TABLE 143 THERMO FISHER SCIENTIFIC INC.: EXPANSIONS, JANUARY 2022-JULY 2025
- TABLE 144 MERCK KGAA: COMPANY OVERVIEW
- TABLE 145 MERCK KGAA: PRODUCTS OFFERED
- TABLE 146 MERCK KGAA: DEALS, JANUARY 2022-DECEMBER 2025
- TABLE 147 MERCK KGAA: EXPANSIONS, JANUARY 2022-DECEMBER 2025
- TABLE 148 MERCK KGAA: OTHER DEVELOPMENTS, JANUARY 2022-DECEMBER 2025
- TABLE 149 SARTORIUS AG COMPANY OVERVIEW
- TABLE 150 SARTORIUS AG: PRODUCTS OFFERED
- TABLE 151 WATERS CORPORATION: COMPANY OVERVIEW
- TABLE 152 WATERS CORPORATION: PRODUCTS OFFERED
- TABLE 153 WATERS CORPORATION: PRODUCT LAUNCHES, JANUARY 2022-DECEMBER 2025
- TABLE 154 WATERS CORPORATION: DEALS, JANUARY 2022-SEPTEMBER 2025
- TABLE 155 AGILENT TECHNOLOGIES, INC.: COMPANY OVERVIEW
- TABLE 156 AGILENT TECHNOLOGIES, INC.: PRODUCTS OFFERED
- TABLE 157 AGILENT TECHNOLOGIES, INC.: PRODUCT LAUNCHES, JANUARY 2022-DECEMBER 2025
- TABLE 158 AGILENT TECHNOLOGIES, INC.: DEALS, JANUARY 2022-DECEMBER 2025
- TABLE 159 AGILENT TECHNOLOGIES, INC.: EXPANSIONS, JANUARY 2022-SEPTEMBER 2025
- TABLE 160 AVANTOR, INC.: COMPANY OVERVIEW
- TABLE 161 AVATOR, INC.: PRODUCTS OFFERED
- TABLE 162 BIO-RAD LABORATORIES, INC.: COMPANY OVERVIEW
- TABLE 163 BIO-RAD LABORATORIES, INC.: PRODUCTS OFFERED
- TABLE 164 REPLIGEN CORPORATION: COMPANY OVERVIEW
- TABLE 165 REPLIGEN CORPORATION: PRODUCTS OFFERED
- TABLE 166 REPLIGEN CORPORATION: PRODUCT LAUNCHES, JANUARY 2022-DECEMBER 2025
- TABLE 167 REPLIGEN CORPORATION: DEALS, JANUARY 2022-DECEMBER 2025
- TABLE 168 SHIMADZU CORPORATION: COMPANY OVERVIEW
- TABLE 169 SHIMADZU CORPORATION: PRODUCTS OFFERED
- TABLE 170 SHIMADZU CORPORATION: DEALS, JANUARY 2022-DECEMBER 2025
- TABLE 171 ECOLAB INC.: COMPANY OVERVIEW
- TABLE 172 ECOLAB INC: PRODUCTS OFFERED
- TABLE 173 ECOLAB INC.: PRODUCT LAUNCHES, JANUARY 2022-DECEMBER 2025
- TABLE 174 ECOLAB INC.: DEALS, JANUARY 2022-DECEMBER 2025
- TABLE 175 ECOLAB INC.: EXPANSIONS, JANUARY 2022-DECEMBER 2025
- TABLE 176 TOSOH BIOSCIENCE: COMPANY OVERVIEW
- TABLE 177 TOSOH BIOSCIENCE: PRODUCTS OFFERED
- TABLE 178 TOSOH BIOSCIENCE: EXPANSIONS, JANUARY 2022-DECEMBER 2025
- TABLE 179 MITSUBISHI CHEMICAL CORPORATION: COMPANY OVERVIEW
- TABLE 180 REVVITY: COMPANY OVERVIEW
- TABLE 181 PERKINELMER: COMPANY OVERVIEW
- TABLE 182 CECIL INSTRUMENTS LIMITED: COMPANY OVERVIEW
- TABLE 183 W. R. GRACE & CO.-CONN: COMPANY OVERVIEW
- TABLE 184 ECOM SPOL. S R.O.: COMPANY OVERVIEW
- TABLE 185 BUCHI LABORTECHNIK AG: COMPANY OVERVIEW
- TABLE 186 GOOD SCIENCE (TIANJIN) INSTRUMENT TECHNOLOGIES CO., LTD.: COMPANY OVERVIEW
- TABLE 187 BIO WORKS TECHNOLOGIES: COMPANY OVERVIEW
- TABLE 188 SYKAM GMBH: COMPANY OVERVIEW
- TABLE 189 SUNRESIN NEW MATERIALS CO. LTD.: COMPANY OVERVIEW
- TABLE 190 YMC CO., LTD.: COMPANY OVERVIEW
- TABLE 191 GENO TECHNOLOGY INC.: COMPANY OVERVIEW
- TABLE 192 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: STUDY ASSUMPTIONS
- TABLE 193 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: RISK ANALYSIS
List of Figures
- FIGURE 1 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET SEGMENTATION & REGIONAL SCOPE
- FIGURE 2 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: YEARS CONSIDERED
- FIGURE 3 MARKET SCENARIO
- FIGURE 4 GLOBAL PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, 2023-2031
- FIGURE 5 MAJOR STRATEGIES ADOPTED BY KEY PLAYERS IN PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, 2024-2025
- FIGURE 6 DISRUPTIONS INFLUENCING GROWTH OF PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET
- FIGURE 7 HIGH-GROWTH SEGMENTS IN PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, 2025-2031
- FIGURE 8 ASIA PACIFIC TO REGISTER HIGHEST CAGR IN PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, IN TERMS OF VALUE, DURING FORECAST PERIOD
- FIGURE 9 INCREASED PRODUCTION OF MONOCLONAL ANTIBODIES TO DRIVE DEMAND FOR BIOPROCESSING AND SEPARATION PRODUCTS
- FIGURE 10 CHINA AND JAPAN TO ACQUIRE A MAJOR SHARE OF MARKET IN ASIA PACIFIC IN 2025
- FIGURE 11 INDIA TO REGISTER HIGHEST CAGR FROM 2026 TO 2030
- FIGURE 12 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: DRIVERS, RESTRAINTS, OPPORTUNITIES, AND CHALLENGES
- FIGURE 13 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: PORTER'S FIVE FORCES ANALYSIS
- FIGURE 14 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: SUPPLY CHAIN ANALYSIS
- FIGURE 15 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: VALUE CHAIN ANALYSIS
- FIGURE 16 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: ECOSYSTEM ANALYSIS
- FIGURE 17 AVERAGE SELLING PRICE OF GC COLUMNS, BY KEY PLAYER, 2025 (USD)
- FIGURE 18 AVERAGE SELLING PRICE OF PREPARATIVE AND PROCESS CHROMATOGRAPHY PRODUCTS, BY REGION, 2025 (USD)
- FIGURE 19 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: IMPORT SCENARIO FOR CHROMATOGRAPHS AND ELECTROPHORESIS INSTRUMENTS (HS CODE 902720)
- FIGURE 20 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: EXPORT SCENARIO FOR CHROMATOGRAPHS AND ELECTROPHORESIS INSTRUMENTS (HS CODE 902720)
- FIGURE 21 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: TRENDS/DISRUPTIONS IMPACTING CUSTOMERS' BUSINESSES
- FIGURE 22 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: INVESTMENT & FUNDING SCENARIO, 2019-2023
- FIGURE 23 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: NUMBER OF INVESTOR DEALS, BY KEY PLAYER, 2019-2023
- FIGURE 24 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: VALUE OF INVESTOR DEALS, BY KEY PLAYER, 2019-2023 (USD MILLION)
- FIGURE 25 PATENT ANALYSIS FOR PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET (JANUARY 2015-JANUARY 2026)
- FIGURE 26 INFLUENCE OF STAKEHOLDERS ON BUYING PROCESS FOR TOP FOUR END USERS
- FIGURE 27 KEY BUYING CRITERIA, BY END USER
- FIGURE 28 NORTH AMERICA: PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET SNAPSHOT (2024)
- FIGURE 29 ASIA PACIFIC: PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET SNAPSHOT (2025)
- FIGURE 30 REVENUE ANALYSIS OF KEY PLAYERS IN PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET, 2020-2024 (USD MILLION)
- FIGURE 31 MARKET SHARE ANALYSIS OF KEY PLAYERS IN PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET (2025)
- FIGURE 32 MARKET RANKING OF KEY PLAYERS IN PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET (2025)
- FIGURE 33 EV/EBITDA OF TOP THREE PLAYERS (2026)
- FIGURE 34 YEAR-TO-DATE (YTD) PRICE TOTAL RETURN AND 5-YEAR STOCK BETA OF TOP THREE PLAYERS (2026)
- FIGURE 35 PREPARATIVE AND PROCESS CHROMATOGRAPHY: BRAND/PRODUCT COMPARATIVE ANALYSIS
- FIGURE 36 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: COMPANY EVALUATION MATRIX (KEY PLAYERS), 2025
- FIGURE 37 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: COMPANY FOOTPRINT
- FIGURE 38 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: COMPANY EVALUATION MATRIX (STARTUPS/SMES), 2025
- FIGURE 39 DANAHER: COMPANY SNAPSHOT (2024)
- FIGURE 40 THERMO FISHER SCIENTIFIC INC.: COMPANY SNAPSHOT (2024)
- FIGURE 41 MERCK KGAA: COMPANY SNAPSHOT (2024)
- FIGURE 42 SARTORIUS AG: COMPANY SNAPSHOT (2024)
- FIGURE 43 WATERS CORPORATION: COMPANY SNAPSHOT (2024)
- FIGURE 44 AGILENT TECHNOLOGIES, INC.: COMPANY SNAPSHOT (2025)
- FIGURE 45 AVANTOR, INC.: COMPANY SNAPSHOT (2024)
- FIGURE 46 BIO-RAD LABORATORIES, INC.: COMPANY SNAPSHOT (2024)
- FIGURE 47 REPLIGEN CORPORATION: COMPANY SNAPSHOT (2024)
- FIGURE 48 SHIMADZU CORPORATION: COMPANY SNAPSHOT (2025)
- FIGURE 49 ECOLAB INC.: COMPANY SNAPSHOT (2024)
- FIGURE 50 TOSOH BIOSCIENCE: COMPANY SNAPSHOT (2024)
- FIGURE 51 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: RESEARCH DATA
- FIGURE 52 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: RESEARCH DESIGN
- FIGURE 53 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: KEY SECONDARY SOURCES
- FIGURE 54 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: KEY DATA FROM SECONDARY SOURCES
- FIGURE 55 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: KEY PRIMARY SOURCES (DEMAND AND SUPPLY SIDES)
- FIGURE 56 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: KEY SUPPLY- AND DEMAND-SIDE PARTICIPANTS
- FIGURE 57 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: BREAKDOWN OF PRIMARY INTERVIEWS (BY COMPANY TYPE, DESIGNATION, AND REGION)
- FIGURE 58 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: KEY INSIGHTS FROM PRIMARY EXPERTS
- FIGURE 59 RESEARCH METHODOLOGY: HYPOTHESIS BUILDING
- FIGURE 60 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: COMPANY REVENUE ESTIMATION
- FIGURE 61 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: END-USER AND REVENUE MAPPING-BASED MARKET SIZE ESTIMATION METHODOLOGY
- FIGURE 62 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: TOP-DOWN APPROACH
- FIGURE 63 GROWTH PROJECTIONS ON REVENUE IMPACT OF KEY MACROINDICATORS
- FIGURE 64 PREPARATIVE AND PROCESS CHROMATOGRAPHY MARKET: DATA TRIANGULATION METHODOLOGY







