 |
市場調查報告書
商品編碼
2019121
去心房顫動市場商業機會、成長要素、產業趨勢分析及2026-2035年預測Defibrillators Market Opportunity, Growth Drivers, Industry Trend Analysis, and Forecast 2026 - 2035 |
||||||
預計到 2025 年,全球去心房顫動市場價值將達到 149 億美元,並預計以 5.5% 的複合年成長率成長,到 2035 年達到 262 億美元。
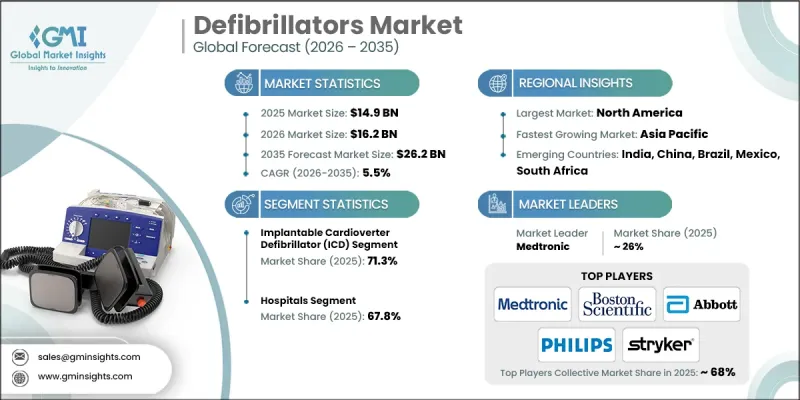
推動市場成長的因素包括心血管疾病盛行率上升、心臟麻痹發生率增加、老年人口成長以及去心房顫動技術的不斷進步。去心房顫動透過可控制的電擊來恢復危及生命的心率患者的正常心律,廣泛應用於醫院、急診室、公共場所,也可作為植入式設備使用。製造商在無線功能、即時病患監測、自動電擊以及與緊急應變系統的整合等領域不斷創新。攜帶式且易於使用的自動體外心臟去心房顫動(AED) 使即使是未經訓練的公眾也能迅速提供救助。穿戴式去心房顫動因其能夠為高風險患者提供持續監測和即時干預、提高患者的行動能力和安全性,以及在植入式解決方案普及之前彌補治療缺口而備受關注。
| 市場範圍 | |
|---|---|
| 開始年份 | 2025 |
| 預測期 | 2026-2035 |
| 上市時的市場規模 | 149億美元 |
| 預測金額 | 262億美元 |
| 複合年成長率 | 5.5% |
預計到2025年,植入式心臟整流去顫器(ICD)市場佔有率將達到71.3%,這主要得益於其在治療危及生命的心律失常方面的廣泛臨床應用、醫生的大力支持以及室性心動過速和室顫病例的增加。 ICD對於長期心臟管理至關重要,因為它可以自動偵測異常心率並進行電擊矯正。
預計到 2025 年,醫院領域將佔據 67.8% 的市場佔有率,達到 180 億美元,這得益於先進去心房顫動的普及、熟練的醫護人員、對複雜心臟病患者的護理以及能夠進行遠端監測和及時警報的數位健康技術。
預計2025年,北美去心房顫動市佔率將達到37.3%,主要促進因素包括心血管疾病高發生率、久坐的生活方式、肥胖以及相關合併症。院外心臟事件的高發生率也推動了對自動體外去顫器(AED)和植入式心臟復律去顫器(ICD)的需求。該地區匯集了許多主要的去心房顫動在全部區域普及和應用。
目錄
第1章:調查方法
- 研究途徑
- 品質改進計劃
- GMI人工智慧政策和資料完整性計劃
- 資訊來源一致性協議
- GMI人工智慧政策和資料完整性計劃
- 調查過程和可靠性評分
- 調查過程的組成部分
- 評分組成部分
- 數據收集
- 主要來源部分列表
- 資料探勘資訊來源
- 付費資訊來源
- 區域資訊來源
- 付費資訊來源
- 基本估算和計算方法
- 每種方法中基準年的計算
- 預測模型
- 量化市場影響分析
- 生長參數對預測的數學影響
- 量化市場影響分析
- 關於調查透明度的補充信息
- 資訊來源歸屬框架
- 品質保證指標
- 對信任的承諾
第2章執行摘要
第3章業界考察
- 生態系分析
- 影響產業的因素
- 促進因素
- 心血管疾病和心臟麻痹的發生率增加
- 植入式和自動去心房顫動的技術進步
- 意識提升對心肺復甦術 (CPR) 和自動體外心臟去心房顫動(AED) 使用的認知,並擴大相關訓練計劃。
- 攜帶式和易於使用的AED設備的廣泛應用
- 產業潛在風險與挑戰
- 去心房顫動設備和維護高成本。
- 產品召回與安全問題
- 市場機遇
- 新興市場除顫去心房顫動使用率不斷提高
- 去心房顫動的研發
- 促進因素
- 成長潛力分析
- 監理情勢(基於初步調查)
- 北美洲
- 歐洲
- 亞太地區
- 科技趨勢
- 當前技術趨勢
- 新興技術
- 還款方案(基於初步調查)
- 未來市場趨勢
- 波特五力分析
- PESTEL 分析
- 客戶洞察(基於初步研究)
- Start-Ups情境(基於初步研究)
- 投資趨勢(基於初步調查)
- 正在研發的產品(基於初步調查)
- 指標趨勢
- 植入式心臟整流去顫器(ICD)
- 穿戴式體外去心房顫動(WCD)
- 價值鏈分析
- 2025年價格分析(基於初步調查)
- 人工智慧的影響及其未來前景
- 差距分析
第4章 競爭情勢
- 介紹
- 企業矩陣分析
- 企業市佔率分析(基於初步研究)
- 世界
- 北美洲
- 歐洲
- 亞太地區
- 競爭定位矩陣
- 主要市場公司的競爭分析
- 主要進展
- 併購
- 夥伴關係與合作
- 新產品發布
- 業務拓展計劃
第5章 市場估價與預測:依產品分類,2022-2035年
- 植入式心臟整流去顫器(ICD)
- 經靜脈植入式心臟整流去顫器
- 雙腔ICD
- 單腔植入式心律轉復除顫器
- 植入式心臟整流去顫器
- 經靜脈植入式心臟整流去顫器
- 體外去心房顫動
- 手動去心房顫動
- 自動ED
- 半自動體外心臟去心房顫動
- 全自動體外心臟去心房顫動
- 去心房顫動
第6章 市場估算與預測:依最終用途分類,2022-2035年
- 醫院
- 門診手術中心
- 其他最終用戶
第7章 市場估計與預測:依地區分類,2022-2035年
- 北美洲
- 美國
- 加拿大
- 歐洲
- 德國
- 英國
- 法國
- 西班牙
- 義大利
- 荷蘭
- 亞太地區
- 中國
- 日本
- 印度
- 澳洲
- 韓國
- 拉丁美洲
- 巴西
- 墨西哥
- 阿根廷
- 中東和非洲
- 南非
- 沙烏地阿拉伯
- 阿拉伯聯合大公國
第8章:公司簡介
- Abbott
- Amiitalia
- Asahi KASEI
- BIOTRONIK
- Boston Scientific
- BPL Medical Technologies
- CU MEDICAL
- MEDITECH
- Medtronic
- MicroPort
- Mindray
- NIHON KOHDEN
- Philips
- SCHILLER
- Stryker
The Global Defibrillators Market was valued at USD 14.9 billion in 2025 and is estimated to grow at a CAGR of 5.5% to reach USD 26.2 billion by 2035.

The market growth is driven by the rising prevalence of cardiovascular diseases, increasing incidents of sudden cardiac arrest, an expanding geriatric population, and continuous advancements in defibrillator technology. Defibrillators deliver controlled electrical shocks to restore normal heart rhythm in life-threatening arrhythmias and are deployed in hospitals, emergency care, public locations, and implantable devices. Manufacturers are innovating with wireless-enabled devices, real-time patient monitoring, automated shock delivery, and integration with emergency response systems. Portable, user-friendly automated external defibrillators (AEDs) allow even untrained bystanders to provide timely assistance. Wearable defibrillators are gaining traction by offering continuous monitoring and immediate intervention for high-risk patients, improving mobility and safety while bridging the gap before implantable solutions.
| Market Scope | |
|---|---|
| Start Year | 2025 |
| Forecast Year | 2026-2035 |
| Start Value | $14.9 Billion |
| Forecast Value | $26.2 Billion |
| CAGR | 5.5% |
The implantable cardioverter defibrillator (ICD) segment held a 71.3% share in 2025, driven by widespread clinical adoption for managing life-threatening arrhythmias, high physician preference, and rising ventricular tachycardia and fibrillation cases. ICDs automatically detect abnormal heart rhythms and deliver corrective shocks, making them essential for long-term cardiac management.
The hospitals segment accounted for 67.8% share in 2025 and is expected to reach USD 18 billion, supported by advanced defibrillator availability, skilled staff, complex cardiac patient care, and digital health technologies enabling remote monitoring and timely alerts.
North America Defibrillators Market held a 37.3% share in 2025 owing to a high incidence of cardiovascular diseases, sedentary lifestyles, obesity, and related comorbidities. The prevalence of out-of-hospital cardiac events has fueled demand for AEDs and ICDs. The region hosts leading defibrillator manufacturers that heavily invest in research, product development, and commercialization. Strategic collaborations with healthcare providers, ongoing product launches, and continuous technological upgrades enhance accessibility and adoption across the region.
Prominent players in the Global Defibrillators Market include Abbott, Amiitalia, Asahi KASEI, BIOTRONIK, Boston Scientific, BPL Medical Technologies, CU MEDICAL, MEDITECH, Medtronic, MicroPort, Mindray, NIHON KOHDEN, Philips, SCHILLER, and Stryker. Companies in the Global Defibrillators Market strengthen their position by investing in R&D for advanced, reliable, and connected devices, expanding product portfolios to include portable, wearable, and implantable solutions. They form partnerships with hospitals, emergency services, and distribution networks to improve reach and adoption. Focused marketing campaigns, clinical validation studies, and training programs for healthcare professionals reinforce product credibility. Regional expansion strategies target high-risk patient populations while maintaining compliance with local regulatory standards. Continuous innovation, cost optimization, and integration of AI-enabled monitoring and telehealth platforms enhance customer satisfaction, brand loyalty, and market foothold.
Table of Contents
Chapter 1 Research Methodology
- 1.1 Research approach
- 1.2 Quality commitments
- 1.2.1 GMI AI policy & data integrity commitment
- 1.2.1.1 Source consistency protocol
- 1.2.1 GMI AI policy & data integrity commitment
- 1.3 Research trail & confidence scoring
- 1.3.1 Research trail components
- 1.3.2 Scoring components
- 1.4 Data collection
- 1.4.1 Partial list of primary sources
- 1.5 Data mining sources
- 1.5.1 Paid sources
- 1.5.1.1 Sources, by region
- 1.5.1 Paid sources
- 1.6 Base estimates and calculations
- 1.6.1 Base year calculation for any one approach
- 1.7 Forecast model
- 1.7.1 Quantified market impact analysis
- 1.7.1.1 Mathematical impact of growth parameters on forecast
- 1.7.1 Quantified market impact analysis
- 1.8 Research transparency addendum
- 1.8.1 Source attribution framework
- 1.8.2 Quality assurance metrics
- 1.8.3 Our commitment to trust
Chapter 2 Executive Summary
- 2.1 Industry 360° synopsis
- 2.2 Key market trends
- 2.2.1 Regional trends
- 2.2.2 Product trends
- 2.2.3 End use trends
- 2.3 CXO perspectives: Strategic imperatives
Chapter 3 Industry Insights
- 3.1 Industry ecosystem analysis
- 3.2 Industry impact forces
- 3.2.1 Growth drivers
- 3.2.1.1 Rising prevalence of cardiovascular diseases and sudden cardiac arrest
- 3.2.1.2 Technological advancements in implantable and automated defibrillators
- 3.2.1.3 Increasing awareness and training programs for CPR and AED usage
- 3.2.1.4 Growing adoption of portable and user-friendly AED devices
- 3.2.2 Industry pitfalls and challenges
- 3.2.2.1 High cost of defibrillator devices and maintenance
- 3.2.2.2 Product recalls and safety concerns
- 3.2.3 Market opportunities
- 3.2.3.1 Expansion of defibrillator deployment in emerging markets
- 3.2.3.2 Development of wearable cardioverter-defibrillators
- 3.2.1 Growth drivers
- 3.3 Growth potential analysis
- 3.4 Regulatory landscape (Driven by primary research)
- 3.4.1 North America
- 3.4.2 Europe
- 3.4.3 Asia Pacific
- 3.5 Technology landscape
- 3.5.1 Current technological trends
- 3.5.2 Emerging technologies
- 3.6 Reimbursement scenario (Driven by primary research)
- 3.7 Future market trends
- 3.8 Porter's analysis
- 3.9 PESTEL analysis
- 3.10 Customer insights (Driven by primary research)
- 3.11 Start-up scenarios (Driven by primary research)
- 3.12 Investment landscape (Driven by primary research)
- 3.13 Pipeline products (Driven by primary research)
- 3.14 Indication landscape
- 3.14.1 Implantable cardioverter defibrillators (ICDs)
- 3.14.2 Wearable external cardioverter defibrillators (WCDs)
- 3.15 Value chain analysis
- 3.16 Pricing analysis, 2025 (Driven by primary research)
- 3.17 Impact of AI and its future assessment
- 3.18 Gap analysis
Chapter 4 Competitive Landscape, 2025
- 4.1 Introduction
- 4.2 Company matrix analysis
- 4.3 Company market share analysis (Driven by primary research)
- 4.3.1 Global
- 4.3.2 North America
- 4.3.3 Europe
- 4.3.4 Asia Pacific
- 4.4 Competitive positioning matrix
- 4.5 Competitive analysis of major market players
- 4.6 Key developments
- 4.6.1 Mergers & acquisitions
- 4.6.2 Partnerships & collaborations
- 4.6.3 New product launches
- 4.6.4 Expansion plans
Chapter 5 Market Estimates and Forecast, By Product, 2022 - 2035 ($ Mn)
- 5.1 Key trends
- 5.2 Implantable cardioverter defibrillator (ICD)
- 5.2.1 Transvenous implantable cardioverter defibrillator
- 5.2.1.1 Dual-chamber ICDs
- 5.2.1.2 Single-chamber ICDs
- 5.2.2 Subcutaneous implantable cardioverter defibrillator
- 5.2.1 Transvenous implantable cardioverter defibrillator
- 5.3 External cardioverter defibrillator
- 5.3.1 Manual ED
- 5.3.2 Automated ED
- 5.3.2.1 Semi-automated external defibrillator
- 5.3.2.2 Fully automated external defibrillator
- 5.3.3 Wearable cardioverter defibrillator
Chapter 6 Market Estimates and Forecast, By End Use, 2022 - 2035 ($ Mn)
- 6.1 Key trends
- 6.2 Hospitals
- 6.3 Ambulatory surgical centers
- 6.4 Other end users
Chapter 7 Market Estimates and Forecast, By Region, 2022 - 2035 ($ Mn)
- 7.1 Key trends
- 7.2 North America
- 7.2.1 U.S.
- 7.2.2 Canada
- 7.3 Europe
- 7.3.1 Germany
- 7.3.2 UK
- 7.3.3 France
- 7.3.4 Spain
- 7.3.5 Italy
- 7.3.6 Netherlands
- 7.4 Asia Pacific
- 7.4.1 China
- 7.4.2 Japan
- 7.4.3 India
- 7.4.4 Australia
- 7.4.5 South Korea
- 7.5 Latin America
- 7.5.1 Brazil
- 7.5.2 Mexico
- 7.5.3 Argentina
- 7.6 Middle East and Africa
- 7.6.1 South Africa
- 7.6.2 Saudi Arabia
- 7.6.3 UAE
Chapter 8 Company Profiles
- 8.1 Abbott
- 8.2 Amiitalia
- 8.3 Asahi KASEI
- 8.4 BIOTRONIK
- 8.5 Boston Scientific
- 8.6 BPL Medical Technologies
- 8.7 CU MEDICAL
- 8.8 MEDITECH
- 8.9 Medtronic
- 8.10 MicroPort
- 8.11 Mindray
- 8.12 NIHON KOHDEN
- 8.13 Philips
- 8.14 SCHILLER
- 8.15 Stryker










