 |
市場調查報告書
商品編碼
1998708
創傷固定裝置市場機會、成長要素、產業趨勢分析及2026-2035年預測Trauma Fixation Devices Market Opportunity, Growth Drivers, Industry Trend Analysis, and Forecast 2026 - 2035 |
||||||
2025 年全球創傷固定裝置市場價值為 96 億美元,預計到 2035 年將達到 134 億美元,年複合成長率為 3.3%。
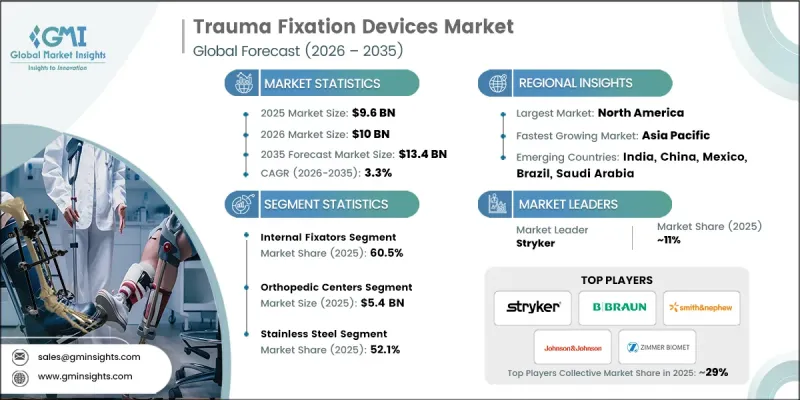
創傷和整形外科損傷發病率的上升、微創手術需求的增加以及內固定技術的不斷進步是推動市場成長的主要因素。創傷固定裝置,包括鋼板、螺絲、鋼棒、鋼針和外固定器,是骨折癒合過程中穩定骨骼的重要植入和器械。交通事故、運動傷害和跌倒的激增,以及易患骨折和骨質疏鬆症的老年人口的成長,都在推動對這些裝置的需求。 3D列印、裝置客製化、智慧技術、物聯網解決方案以及鈦和其他先進材料的應用等趨勢,進一步改善了臨床療效和手術精度,推動了整體市場擴張,並塑造著全球整形外科醫學的未來。
| 市場範圍 | |
|---|---|
| 開始年份 | 2025 |
| 預測期 | 2026-2035 |
| 上市時的市場規模 | 96億美元 |
| 預測金額 | 134億美元 |
| 複合年成長率 | 3.3% |
2025年,內固定裝置器械將佔60.5%的市場。這主要得益於其能夠穩定複雜骨折、提供卓越的動態強度並支持精確的解剖復位。髓內釘、鋼板和螺絲等器械能夠實現微創手術,減少肌肉和組織的損傷,加速康復,並確保骨骼的穩定癒合。
到2025年,不銹鋼市佔率將達到52.1%。與鈦相比,不銹鋼價格更實惠,因此在注重成本的醫療機構和新興市場中,它成為一種易於獲取的選擇。此外,不銹鋼的高抗張強度和耐久性也推動了其在全球整形外科領域的應用,用於支撐鋼板、螺絲和連接桿等高負荷應用。
預計到2025年,北美創傷固定器材市場將佔據54.8%的佔有率,這主要得益於該地區高發生率的創傷病例、先進的醫療基礎設施和專業的整形外科中心。骨折的高發生率,加上先進固定器械的普及,正在推動區域需求成長,並改善病患的治療效果。
目錄
第1章:調查方法
- 研究途徑
- 品質改進計劃
- GMI人工智慧政策和資料完整性計劃
- 資訊來源一致性通訊協定
- GMI人工智慧政策和資料完整性計劃
- 調查過程和可靠性評分
- 調查過程的組成部分
- 評分組成部分
- 數據收集
- 主要來源部分列表
- 資料探勘資訊來源
- 付費資訊來源
- 區域資訊來源
- 付費資訊來源
- 基本估算和計算方法
- 每種方法中基準年的計算
- 預測模型
- 量化市場影響分析
- 生長參數對預測的數學影響
- 量化市場影響分析
- 關於調查透明度的補充信息
- 資訊來源歸屬框架
- 品質保證指標
- 對信任的承諾
第2章執行摘要
第3章業界考察
- 生態系分析
- 影響產業的因素
- 促進因素
- 退化性骨病率增加
- 受傷人數增加
- 創傷固定裝置的技術進步
- 對微創手術的需求日益成長
- 產業潛在風險與挑戰
- 術後併發症
- 嚴格的規定
- 市場機遇
- 擴大研發投資及產品開發活動
- 促進因素
- 成長潛力分析
- 監理情勢
- 北美洲
- 歐洲
- 亞太地區
- 科技趨勢
- 當前技術趨勢
- 新興技術
- 未來市場趨勢
- 2022-2025年各地區道路交通事故死亡人數
- 管道分析
- 專利分析
- 價格分析(2025 年)(基於初步調查)
- 客戶洞察(基於初步研究)
- 波特五力分析
- PESTEL 分析
- 差距分析
第4章 競爭情勢
- 介紹
- 企業矩陣分析
- 企業市佔率分析
- 世界
- 北美洲
- 歐洲
- 亞太地區
- LAMEA
- 競爭定位矩陣
- 主要市場公司的競爭分析
- 主要進展
- 併購
- 夥伴關係與合作
- 新產品類型發布
- 業務拓展計劃
第5章 市場估價與預測:依產品分類,2022-2035年
- 內部固定裝置
- 盤子
- 指甲
- 擰緊
- 其他內部固定裝置
- 外固定器
- 單側和雙側
- 混合
- 圓
第6章 市場估算與預測:依材料分類 2022-2035
- 不銹鋼
- 鈦
- 其他材料
第7章 市場估價與預測:依零件分類,2022-2035年
- 下肢
- 腳和腳踝
- 膝蓋
- 小腿
- 髖關節和骨盆
- 大腿
- 上肢
- 上肢
- 手腕和手
- 肩膀
- 彎頭
第8章 市場估算與預測:依最終用途分類,2022-2035年
- 整形外科中心
- 醫院
- 門診手術中心
- 其他最終用戶
第9章 市場估計與預測:依地區分類,2022-2035年
- 北美洲
- 美國
- 加拿大
- 歐洲
- 德國
- 英國
- 法國
- 西班牙
- 義大利
- 荷蘭
- 亞太地區
- 中國
- 日本
- 印度
- 澳洲
- 韓國
- 拉丁美洲
- 巴西
- 墨西哥
- 阿根廷
- 中東和非洲
- 南非
- 沙烏地阿拉伯
- 阿拉伯聯合大公國
第10章:公司簡介
- Acumed
- Arthrex
- B Braun
- Bioretec
- CONMED
- Implanet
- Integra
- Johnson &Johnson
- KLS Martin
- Medicon
- Orthofix
- Smith+Nephew
- Stryker
- Wright Medical
- Zimmer Biomet
The Global Trauma Fixation Devices Market was valued at USD 9.6 billion in 2025 and is estimated to grow at a CAGR of 3.3% to reach USD 13.4 billion by 2035.

Market growth is driven by the rising prevalence of trauma and orthopedic injuries, increasing demand for minimally invasive procedures, and ongoing advancements in internal fixation technologies. Trauma fixation devices are essential implants and instruments that stabilize fractured bones during the healing process, including plates, screws, rods, pins, and external fixators. The surge in road accidents, sports-related injuries, and falls, combined with a growing elderly population prone to fractures and osteoporosis, is intensifying the need for these devices. Trends such as the integration of 3D printing, device customization, smart technology, IoT-enabled solutions, and the use of titanium and other advanced materials are further enhancing clinical outcomes, surgical precision, and overall market expansion, shaping the future of orthopedic care globally.
| Market Scope | |
|---|---|
| Start Year | 2025 |
| Forecast Year | 2026-2035 |
| Start Value | $9.6 Billion |
| Forecast Value | $13.4 Billion |
| CAGR | 3.3% |
The internal fixators segment held a share of 60.5% in 2025, driven by their ability to stabilize complex fractures, deliver superior biomechanical strength, and support precise anatomical alignment. Devices such as intramedullary nails, plates, and screws allow minimally invasive approaches that reduce muscle and tissue trauma, accelerate recovery, and ensure consistent bone healing.
The stainless steel segment held a 52.1% share in 2025. Its affordability compared to titanium makes it an accessible option for cost-sensitive healthcare settings and emerging markets. It also provides high tensile strength and durability, supporting plates, screws, and rods in high-load-bearing applications, thereby enhancing adoption across global orthopedic facilities.
North America Trauma Fixation Devices Market held a 54.8% share in 2025, fueled by a high incidence of trauma cases, advanced healthcare infrastructure, and specialized orthopedic centers. The prevalence of fractures, coupled with access to technologically advanced fixation devices, ensures better patient outcomes, driving regional demand.
Prominent players in the Global Trauma Fixation Devices Market include Acumed, Arthrex, B Braun, Bioretec, CONMED, Implanet, Integra, Johnson & Johnson, KLS Martin, Medicon, Orthofix, Smith & Nephew, Stryker, Wright Medical, and Zimmer Biomet. Key strategies adopted by companies to strengthen their Trauma Fixation Devices Market presence include investing in R&D to develop next-generation internal and external fixation solutions, launching minimally invasive and customizable devices, leveraging 3D printing for patient-specific implants, expanding into emerging markets, forming partnerships with hospitals and orthopedic centers, enhancing supply chain efficiency, integrating IoT-enabled and smart technologies for real-time monitoring, providing comprehensive surgeon training and support programs, and focusing on cost-effective stainless steel and titanium-based products to capture both premium and value-driven segments. These approaches enable sustained growth, technological leadership, and improved adoption across global trauma care markets.
Table of Contents
Chapter 1 Research Methodology
- 1.1 Research approach
- 1.2 Quality commitments
- 1.2.1 GMI AI policy & data integrity commitment
- 1.2.1.1 Source consistency protocol
- 1.2.1 GMI AI policy & data integrity commitment
- 1.3 Research trail & confidence scoring
- 1.3.1 Research trail components
- 1.3.2 Scoring components
- 1.4 Data collection
- 1.4.1 Partial list of primary sources
- 1.5 Data mining sources
- 1.5.1 Paid sources
- 1.5.1.1 Sources, by region
- 1.5.1 Paid sources
- 1.6 Base estimates and calculations
- 1.6.1 Base year calculation for any one approach
- 1.7 Forecast model
- 1.7.1 Quantified market impact analysis
- 1.7.1.1 Mathematical impact of growth parameters on forecast
- 1.7.1 Quantified market impact analysis
- 1.8 Research transparency addendum
- 1.8.1 Source attribution framework
- 1.8.2 Quality assurance metrics
- 1.8.3 Our commitment to trust
Chapter 2 Executive Summary
- 2.1 Industry 3600 synopsis
- 2.2 Key market trends
- 2.2.1 Regional trends
- 2.2.2 Product trends
- 2.2.3 Material trends
- 2.2.4 Site trends
- 2.2.5 End use trends
- 2.3 CXO perspectives: Strategic imperatives
Chapter 3 Industry Insights
- 3.1 Industry ecosystem analysis
- 3.2 Industry impact forces
- 3.2.1 Growth drivers
- 3.2.1.1 Increasing prevalence of degenerative bone diseases
- 3.2.1.2 Rising incidence of injuries
- 3.2.1.3 Growing technological advancements in trauma fixation devices
- 3.2.1.4 Increasing demand for minimally invasive procedures
- 3.2.2 Industry pitfalls and challenges
- 3.2.2.1 Post-surgery complication
- 3.2.2.2 Stringent regulation
- 3.2.3 Market opportunities
- 3.2.3.1 Increasing R&D investment and product-development activities
- 3.2.1 Growth drivers
- 3.3 Growth potential analysis
- 3.4 Regulatory landscape
- 3.4.1 North America
- 3.4.2 Europe
- 3.4.3 Asia Pacific
- 3.5 Technology landscape
- 3.5.1 Current technological trends
- 3.5.2 Emerging technologies
- 3.6 Future market trends
- 3.7 Number of fatalities in road accidents, by region, 2022-2025
- 3.7.1 North America
- 3.7.2 Europe
- 3.7.3 Asia Pacific
- 3.7.4 Latin America
- 3.7.5 MEA
- 3.8 Pipeline analysis
- 3.9 Patent analysis
- 3.10 Pricing analysis, 2025 (Driven by primary research)
- 3.11 Customer insights (Driven by primary research)
- 3.12 Porter's analysis
- 3.13 PESTEL analysis
- 3.14 Gap analysis
Chapter 4 Competitive Landscape, 2025
- 4.1 Introduction
- 4.2 Company matrix analysis
- 4.3 Company market share analysis
- 4.3.1 Global
- 4.3.2 North America
- 4.3.3 Europe
- 4.3.4 Asia Pacific
- 4.3.5 LAMEA
- 4.4 Competitive positioning matrix
- 4.5 Competitive analysis of major market players
- 4.6 Key developments
- 4.6.1 Mergers & acquisitions
- 4.6.2 Partnerships & collaborations
- 4.6.3 New product type launches
- 4.6.4 Expansion plans
Chapter 5 Market Estimates and Forecast, By Product, 2022 - 2035 ($ Mn)
- 5.1 Key trends
- 5.2 Internal fixators
- 5.2.1 Plates
- 5.2.2 Nails
- 5.2.3 Screws
- 5.2.4 Other internal fixators
- 5.3 External fixators
- 5.3.1 Unilateral and bilateral
- 5.3.2 Hybrid
- 5.3.3 Circular
Chapter 6 Market Estimates and Forecast, By Material 2022 - 2035 ($ Mn)
- 6.1 Key trends
- 6.2 Stainless steel
- 6.3 Titanium
- 6.4 Other materials
Chapter 7 Market Estimates and Forecast, By Site, 2022 - 2035 ($ Mn)
- 7.1 Key trends
- 7.2 Lower extremities
- 7.2.1 Foot & ankle
- 7.2.2 Knee
- 7.2.3 Lower leg
- 7.2.4 Hip and pelvic
- 7.2.5 Thigh
- 7.3 Upper extremities
- 7.3.1 Arm
- 7.3.2 Hand & wrist
- 7.3.3 Shoulder
- 7.3.4 Elbow
Chapter 8 Market Estimates and Forecast, By End Use, 2022 - 2035 ($ Mn)
- 8.1 Key trends
- 8.2 Orthopedic centers
- 8.3 Hospitals
- 8.4 Ambulatory surgical centers
- 8.5 Other end users
Chapter 9 Market Estimates and Forecast, By Region, 2022 - 2035 ($ Mn)
- 9.1 Key trends
- 9.2 North America
- 9.2.1 U.S.
- 9.2.2 Canada
- 9.3 Europe
- 9.3.1 Germany
- 9.3.2 UK
- 9.3.3 France
- 9.3.4 Spain
- 9.3.5 Italy
- 9.3.6 Netherlands
- 9.4 Asia Pacific
- 9.4.1 China
- 9.4.2 Japan
- 9.4.3 India
- 9.4.4 Australia
- 9.4.5 South Korea
- 9.5 Latin America
- 9.5.1 Brazil
- 9.5.2 Mexico
- 9.5.3 Argentina
- 9.6 Middle East and Africa
- 9.6.1 South Africa
- 9.6.2 Saudi Arabia
- 9.6.3 UAE
Chapter 10 Company Profiles
- 10.1 Acumed
- 10.2 Arthrex
- 10.3 B Braun
- 10.4 Bioretec
- 10.5 CONMED
- 10.6 Implanet
- 10.7 Integra
- 10.8 Johnson & Johnson
- 10.9 KLS Martin
- 10.10 Medicon
- 10.11 Orthofix
- 10.12 Smith+Nephew
- 10.13 Stryker
- 10.14 Wright Medical
- 10.15 Zimmer Biomet








