|
市場調查報告書
商品編碼
1936599
肝癌藥物市場機會、成長要素、產業趨勢分析及2026年至2035年預測Liver Cancer Drugs Market Opportunity, Growth Drivers, Industry Trend Analysis, and Forecast 2026 - 2035 |
||||||
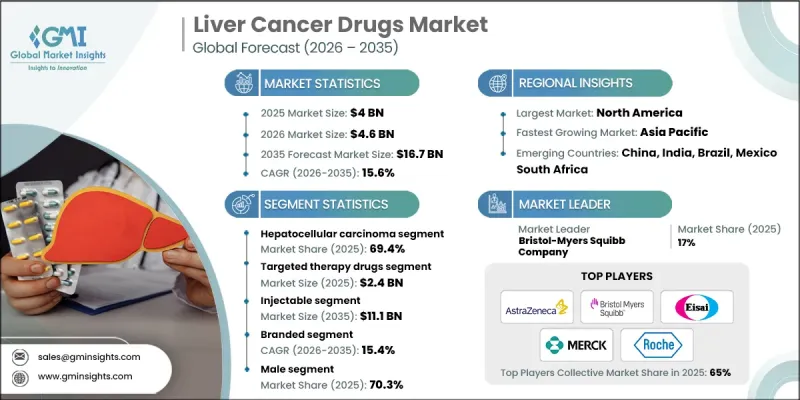
全球肝癌治療市場預計到 2025 年將達到 40 億美元,到 2035 年將達到 167 億美元,年複合成長率為 15.6%。
全球原發性肝癌發病率不斷上升,以及因診斷延遲和根治性選擇有限而導致的高死亡率,推動了市場成長。肝癌仍然是最致命的腫瘤疾病之一,因此迫切需要有效的藥物療法來延長存活期並延緩疾病進展。肝癌治療市場涵蓋用於治療肝臟惡性腫瘤的藥物的研發、生產和商業化,包括旨在改善治療效果和生活品質的療法。藥物研發的重點正日益轉向標靶治療、免疫療法、生物製藥和化療方案。由於許多患者在確診時已處於晚期,全身性治療在疾病管理中發揮關鍵作用。強大的腫瘤學研發管線、支持性的法規結構以及不斷擴大的醫保覆蓋範圍,持續促進創新並加速病患獲得治療。隨著精準醫療和聯合治療策略的興起,對肝癌治療的製藥投資不斷增加,為市場的持續長期成長奠定了基礎。
| 市場覆蓋範圍 | |
|---|---|
| 開始年份 | 2025 |
| 預測年份 | 2026-2035 |
| 起始值 | 40億美元 |
| 預測金額 | 167億美元 |
| 複合年成長率 | 15.6% |
2025年,肝細胞癌細分市場佔據了69.4%的市場佔有率,預計到2035年將以15.5%的複合年成長率成長。這一主導地位反映了該癌症類型在全球範圍內的高發病率以及治療方法(包括聯合治療和免疫療法)的不斷進步。基礎肝病的持續高發生率也持續支撐著對有效治療方案的強勁需求。
預計到2025年,注射劑市場規模將達到27億美元,到2035年將成長至111億美元。注射劑製劑廣泛應用於高級醫療機構,因為它們非常適合需要嚴格控制給藥和臨床監測的免疫療法和生物製藥。注射劑在一線療法和聯合治療中的既定地位進一步鞏固了其市場主導地位。
預計到2025年,北美肝癌治療市場將佔據41.1%的市場。這一區域主導地位得益於健全的監管體系、先進的診斷基礎設施、創新治療方法的早期應用以及完善的臨床研究生態系統。有利的醫保報銷政策以及對早期檢測和治療最佳化的日益重視,將繼續推動市場需求。
目錄
第1章調查方法和範圍
第2章執行摘要
第3章業界考察
- 生態系分析
- 產業影響因素
- 促進要素
- 全球肝癌發生率不斷上升
- 轉向標靶治療和免疫腫瘤療法
- 老齡人口的增加
- 醫療保健成本不斷上漲以及對腫瘤學的關注
- 產業潛在風險與挑戰
- 高昂的治療費用和報銷限制
- 診斷延遲會限制治療合格。
- 市場機遇
- 下一代聯合治療治療和個人化治療
- 新興和高負擔地區的成長潛力
- 促進要素
- 成長潛力分析
- 監管環境
- 北美洲
- 歐洲
- 亞太地區
- 各地區肝癌統計數據
- 未來市場趨勢
- 管道分析
- 波特五力分析
- PESTEL 分析
第4章 競爭情勢
- 介紹
- 公司市佔率分析
- 世界
- 北美洲
- 歐洲
- 亞太地區
- 企業矩陣分析
- 主要市場公司的競爭分析
- 競爭定位矩陣
- 重大進展
- 併購
- 夥伴關係與合作
- 新產品發布
- 擴張計劃
第5章 按類型分類的市場估算與預測,2022-2035年
- 肝細胞癌
- 膽管癌
- 肝母細胞瘤
- 肝轉移
- 其他類型
6. 2022-2035年按藥物類別分類的市場估算與預測
- 化療藥物
- 標靶治療藥物
- 免疫療法
7. 依行政途徑分類的市場估計與預測,2022-2035 年
- 口服
- 注射
8. 按藥物類型分類的市場估計和預測,2022-2035 年
- 學名藥
- 品牌產品
第9章 依性別分類的市場估計與預測,2022-2035年
- 男性
- 女士
第10章 依最終用途分類的市場估計與預測,2022-2035年
- 醫院
- 專科癌症中心
- 研究和學術機構
第11章 2022-2035年各地區市場估計與預測
- 北美洲
- 美國
- 加拿大
- 歐洲
- 德國
- 英國
- 法國
- 西班牙
- 義大利
- 荷蘭
- 亞太地區
- 中國
- 印度
- 日本
- 澳洲
- 韓國
- 拉丁美洲
- 巴西
- 墨西哥
- 阿根廷
- 中東和非洲
- 南非
- 沙烏地阿拉伯
- 阿拉伯聯合大公國
第12章:公司簡介
- AbbVie
- Amgen
- AstraZeneca
- Bayer
- Bristol-Myers Squibb Company
- Eisai
- Exelixis
- F. Hoffmann-La Roche
- Glenmark Pharmaceuticals
- Johnson &Johnson
- Merck &Co.
- Regeneron Pharmaceuticals
- Sanofi
- Servier Pharmaceuticals
- Taiho Pharmaceutical
The Global Liver Cancer Drugs Market was valued at USD 4 billion in 2025 and is estimated to grow at a CAGR of 15.6% to reach USD 16.7 billion by 2035.

Market growth is driven by the rising incidence of primary liver cancers worldwide and the persistently high mortality rates associated with delayed diagnosis and limited curative treatment options. Liver cancer remains one of the most fatal oncology indications, creating an urgent need for effective drug-based therapies that can extend survival and slow disease progression. The liver cancer drugs market encompasses the research, production, and commercialization of pharmaceutical treatments used to manage liver malignancies, including therapies designed to improve outcomes and quality of life. Drug innovation is increasingly focused on targeted agents, immunotherapies, biologics, and chemotherapy-based regimens. Because many patients are diagnosed at advanced stages, systemic therapies play a critical role in disease management. Strong oncology research pipelines, supportive regulatory frameworks, and expanding reimbursement coverage continue to encourage innovation and accelerate patient access. As precision medicine and combination treatment strategies gain traction, pharmaceutical investment in liver cancer therapeutics is intensifying, positioning the market for sustained long-term expansion.
| Market Scope | |
|---|---|
| Start Year | 2025 |
| Forecast Year | 2026-2035 |
| Start Value | $4 Billion |
| Forecast Value | $16.7 Billion |
| CAGR | 15.6% |
The hepatocellular carcinoma segment accounted for 69.4% share in 2025 and is expected to grow at a CAGR of 15.5% throughout 2035. This dominance reflects the high global burden of this cancer type and continued advancements in treatment approaches, including combination therapies and immune-based drugs. Ongoing prevalence of underlying liver conditions continues to support strong demand for effective therapeutic options.
The injectable segment generated USD 2.7 billion in 2025 and is projected to grow to USD 11.1 billion by 2035. Injectable formulations are widely used in advanced treatment settings due to their suitability for immunotherapies and biologics, which require controlled administration and clinical supervision. Their established role in first line and combination regimens further supports segment leadership.
North America Liver Cancer Drugs Market held 41.1% share in 2025. Regional dominance is supported by strong regulatory pathways, advanced diagnostic infrastructure, early adoption of innovative therapies, and a well-developed clinical research ecosystem. Favorable reimbursement policies and increasing focus on early detection and treatment optimization continue to drive demand.
Key companies operating in the Global Liver Cancer Drugs Market include Merck & Co., F. Hoffmann-La Roche, AstraZeneca, Bayer, Bristol-Myers Squibb Company, Regeneron Pharmaceuticals, Johnson & Johnson, Eisai, Exelixis, Sanofi, Amgen, AbbVie, Taiho Pharmaceutical, Servier Pharmaceuticals, and Glenmark Pharmaceuticals. Companies in the liver cancer drugs market are strengthening their competitive position through aggressive research and development initiatives focused on novel drug targets and combination therapies. Many players are expanding immuno-oncology portfolios and investing in precision medicine to improve treatment response rates. Strategic collaborations with research institutions and biotechnology firms are accelerating clinical development timelines. Companies are also prioritizing regulatory approvals across multiple regions to expand market access. Lifecycle management strategies, including label expansions and next-generation formulations, are being used to extend product value.
Table of Contents
Chapter 1 Methodology and Scope
- 1.1 Market scope and definition
- 1.2 Research approach
- 1.3 Quality commitments
- 1.3.1 GMI AI policy and data integrity commitment
- 1.3.1.1 Source consistency protocol
- 1.3.1 GMI AI policy and data integrity commitment
- 1.4 Research trail and confidence scoring
- 1.4.1 Research trail components
- 1.4.2 Scoring components
- 1.5 Data collection
- 1.5.1 Partial list of primary sources
- 1.6 Data mining sources
- 1.6.1 Paid sources
- 1.6.1.1 Sources, by region
- 1.6.1 Paid sources
- 1.7 Base estimates and calculations
- 1.7.1 Revenue share analysis
- 1.7.2 Base year calculation
- 1.8 Forecast model
- 1.9 Research transparency addendum
- 1.9.1 Source attribution framework
- 1.9.2 Quality assurance metrics
- 1.9.3 Our commitment to trust
Chapter 2 Executive Summary
- 2.1 Industry 360° synopsis
- 2.2 Key market trends
- 2.2.1 Regional trends
- 2.2.2 Type trends
- 2.2.3 Drug class trends
- 2.2.4 Route of administration trends
- 2.2.5 Medication type trends
- 2.2.6 Gender trends
- 2.2.7 Distribution channel trends
- 2.3 CXO perspectives: Strategic imperatives
- 2.3.1 Key decision points for industry executives
- 2.3.2 Critical success factors for market players
- 2.4 Future outlook and strategic recommendations
Chapter 3 Industry Insights
- 3.1 Industry ecosystem analysis
- 3.2 Industry impact forces
- 3.2.1 Growth drivers
- 3.2.1.1 Rising global incidence of liver cancer
- 3.2.1.2 Shift toward targeted and immuno-oncology therapies
- 3.2.1.3 Expanding geriatric population
- 3.2.1.4 Rising healthcare expenditure and oncology focus
- 3.2.2 Industry pitfalls and challenges
- 3.2.2.1 High treatment costs and reimbursement constraints
- 3.2.2.2 Late diagnosis limiting treatment eligibility
- 3.2.3 Market opportunities
- 3.2.3.1 Next-generation combination and personalized therapies
- 3.2.3.2 Growth potential in emerging, high-burden regions
- 3.2.1 Growth drivers
- 3.3 Growth potential analysis
- 3.4 Regulatory landscape
- 3.4.1 North America
- 3.4.2 Europe
- 3.4.3 Asia Pacific
- 3.5 Liver cancer statistics, by region
- 3.6 Future market trends
- 3.7 Pipeline analysis
- 3.8 Porter's analysis
- 3.9 PESTEL analysis
Chapter 4 Competitive Landscape, 2025
- 4.1 Introduction
- 4.2 Company market share analysis
- 4.2.1 Global
- 4.2.2 North America
- 4.2.3 Europe
- 4.2.4 Asia Pacific
- 4.3 Company matrix analysis
- 4.4 Competitive analysis of major market players
- 4.5 Competitive positioning matrix
- 4.6 Key developments
- 4.6.1 Merger and acquisition
- 4.6.2 Partnership and collaboration
- 4.6.3 New product launches
- 4.6.4 Expansion plans
Chapter 5 Market Estimates and Forecast, By Type, 2022 - 2035 ($ Mn)
- 5.1 Key trends
- 5.2 Hepatocellular carcinoma
- 5.3 Cholangiocarcinoma
- 5.4 Hepatoblastoma
- 5.5 Liver metastasis
- 5.6 Other types
Chapter 6 Market Estimates and Forecast, By Drug Class, 2022 - 2035 ($ Mn)
- 6.1 Key trends
- 6.2 Chemotherapeutic agents
- 6.3 Targeted therapy drugs
- 6.4 Immunotherapy drugs
Chapter 7 Market Estimates and Forecast, By Route of Administration, 2022 - 2035 ($ Mn)
- 7.1 Key trends
- 7.2 Oral
- 7.3 Injectable
Chapter 8 Market Estimates and Forecast, By Medication Type, 2022 - 2035 ($ Mn)
- 8.1 Key trends
- 8.2 Generic
- 8.3 Branded
Chapter 9 Market Estimates and Forecast, By Gender, 2022 - 2035 ($ Mn)
- 9.1 Key trends
- 9.2 Male
- 9.3 Female
Chapter 10 Market Estimates and Forecast, By End Use, 2022 - 2035 ($ Mn)
- 10.1 Key trends
- 10.2 Hospitals
- 10.3 Specialty cancer centers
- 10.4 Research and academic centers
Chapter 11 Market Estimates and Forecast, By Region, 2022 - 2035 ($ Mn)
- 11.1 Key trends
- 11.2 North America
- 11.2.1 U.S.
- 11.2.2 Canada
- 11.3 Europe
- 11.3.1 Germany
- 11.3.2 UK
- 11.3.3 France
- 11.3.4 Spain
- 11.3.5 Italy
- 11.3.6 Netherlands
- 11.4 Asia Pacific
- 11.4.1 China
- 11.4.2 India
- 11.4.3 Japan
- 11.4.4 Australia
- 11.4.5 South Korea
- 11.5 Latin America
- 11.5.1 Brazil
- 11.5.2 Mexico
- 11.5.3 Argentina
- 11.6 Middle East and Africa
- 11.6.1 South Africa
- 11.6.2 Saudi Arabia
- 11.6.3 UAE
Chapter 12 Company Profiles
- 12.1 AbbVie
- 12.2 Amgen
- 12.3 AstraZeneca
- 12.4 Bayer
- 12.5 Bristol-Myers Squibb Company
- 12.6 Eisai
- 12.7 Exelixis
- 12.8 F. Hoffmann-La Roche
- 12.9 Glenmark Pharmaceuticals
- 12.10 Johnson & Johnson
- 12.11 Merck & Co.
- 12.12 Regeneron Pharmaceuticals
- 12.13 Sanofi
- 12.14 Servier Pharmaceuticals
- 12.15 Taiho Pharmaceutical