 |
市場調查報告書
商品編碼
1797739
前列腺癌生物標記市場機會、成長動力、產業趨勢分析及 2025 - 2034 年預測Prostate Cancer Biomarkers Market Opportunity, Growth Drivers, Industry Trend Analysis, and Forecast 2025 - 2034 |
||||||
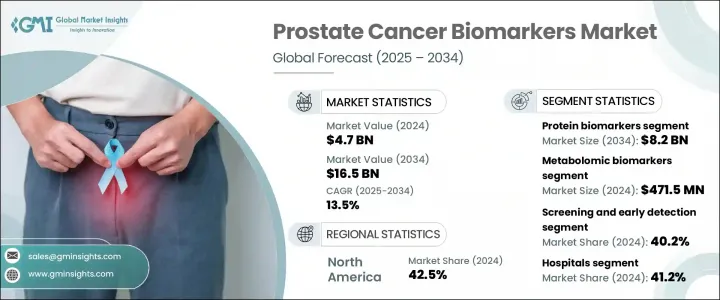
2024 年全球前列腺癌生物標記市場價值為 47 億美元,預計到 2034 年將以 13.5% 的複合年成長率成長至 165 億美元。市場成長主要得益於前列腺癌病例數量的增加(尤其是在老化男性群體中),以及先進診斷技術的進步。人們對早期篩檢的認知不斷提高,加上政府資助的醫療保健計劃的增加,推動了初級保健機構對前列腺癌生物標記的採用。人工智慧驅動的評估、液體活體組織切片和基因組分析平台等下一代診斷技術的整合,正在增強早期檢測能力,同時支援個人化治療決策。新興經濟體也在增加對衛生基礎設施的投資,這正在擴大精準檢測方法的覆蓋範圍。這些生物標記驅動的策略正在透過提供更高的準確性、更低的侵入性和更快的診斷工作流程來重塑癌症治療。
OPKO Health、貝克曼庫爾特、Bio-Rad Laboratories 和 Bio-Techne 等行業主要參與者正引領前列腺癌生物標記市場的創新。這些公司不僅透過尖端診斷平台提高檢測的準確性和速度,更致力於塑造個人化癌症治療的未來。透過開發非侵入性、高靈敏度的生物標記檢測方法,並將其與基於人工智慧的診斷系統相結合,它們能夠更早地識別疾病,更好地監測病情進展,並制定更有效的治療方案。
| 市場範圍 | |
|---|---|
| 起始年份 | 2024 |
| 預測年份 | 2025-2034 |
| 起始值 | 47億美元 |
| 預測值 | 165億美元 |
| 複合年成長率 | 13.5% |
蛋白質生物標記細分市場在2024年創收24億美元,預計2034年將達到82億美元,複合年成長率為13.1%。其主導地位源自於廣泛的臨床認可及其在疾病檢測和進展追蹤中的常規應用。此類生物標記易於透過標準血液檢測進行測量,由於其可靠性、成本效益以及與前列腺癌風險的強相關性,仍然是一線診斷工具。正在進行的研究正在不斷改進這些生物標記技術,使其在保持準確性的同時減少不必要的侵入性診斷。新的基於生物標記的評分工具也在改善風險分層和患者選擇,以便進行進一步檢測,從而最佳化治療途徑。
在應用方面,篩檢和早期檢測領域在2024年佔據了最高的市場佔有率,達到40.2%。這歸因於公共衛生系統和個別臨床實踐對早期診斷的日益重視。如今,篩檢平台專為高容量工作流程而設計,利用血液或尿液生物標記檢測,以最少的處理流程提供快速可靠的結果。這些解決方案非常適合常規健康檢查,並且在快速週轉和易用性至關重要的門診環境中迅速普及。非侵入性檢測的便利性和可近性使其成為全球前列腺癌預防策略中不可或缺的一部分。
2024年,北美前列腺癌生物標記市場佔據42.5%的市場。該地區受益於先進的醫療基礎設施、較高的發病率以及加快的法規核准程序。在強勁的醫療支出和不斷加強的公眾意識宣傳活動的推動下,美國和加拿大在採用尖端診斷平台方面繼續保持領先地位。持續的研究投入、完善的診斷生態系統以及主要市場參與者的存在,進一步支持了該地區的創新。隨著對可靠、快速和微創癌症診斷的需求不斷成長,預計這一領先地位將持續下去。
Myriad Genetics、QIAGEN、西門子、Veracyte、Genomic Health、羅氏製藥、MDxHealth、賽默飛世爾科技和默克集團等公司是重塑競爭格局的關鍵參與者。這些公司透過研發投資擴大檢測組合,並加強與腫瘤實驗室、學術機構和醫院的合作。這些公司正在利用先進的基因組學和蛋白質組學技術來提高其生物標記平台的靈敏度和特異性。他們也進行策略合作,以促進全球分銷、擴大生產規模並獲得監管部門的批准,從而加快產品上市速度。許多公司專注於伴隨診斷的開發,這與精準醫療直接相關,有助於增強治療方案的一致性和患者預後。
目錄
第1章:方法論與範圍
第2章:執行摘要
第3章:行業洞察
- 產業生態系統分析
- 產業衝擊力
- 成長動力
- 攝護腺癌發生率上升
- 基於生物標記的診斷技術的進展
- 公營和私營部門增加研發投資
- 透過生物標記檢測實現個人化醫療的日益普及
- 產業陷阱與挑戰
- 進階診斷測試成本高昂
- 農村和低度開發地區的取得途徑和認知度有限
- 市場機會
- 非侵入性生物標記平台投資不斷增加
- 擴大使用組合生物標記組進行早期診斷
- 成長動力
- 成長潛力分析
- 監管格局
- 技術進步
- 當前的技術趨勢
- 新興技術
- 未來市場趨勢
- 差距分析
- 波特的分析
- PESTEL分析
第4章:競爭格局
- 介紹
- 公司市佔率分析
- 公司矩陣分析
- 主要市場參與者的競爭分析
- 競爭定位矩陣
- 關鍵進展
- 併購
- 夥伴關係和合作
- 新產品發布
- 擴張計劃
第5章:市場估計與預測:按類型,2021 - 2034 年
- 主要趨勢
- 基因生物標記
- 蛋白質生物標記
- 基於細胞的生物標記
- 代謝組學生物標記
第6章:市場估計與預測:按應用,2021 - 2034 年
- 主要趨勢
- 篩檢和早期檢測
- 診斷和風險分層
- 預後和治療監測
- 伴隨診斷
第7章:市場估計與預測:依最終用途,2021 - 2034 年
- 主要趨勢
- 醫院
- 診斷實驗室
- 癌症研究機構
- 生物製藥公司
第8章:市場估計與預測:按地區,2021 - 2034 年
- 主要趨勢
- 北美洲
- 美國
- 加拿大
- 歐洲
- 德國
- 英國
- 法國
- 西班牙
- 義大利
- 荷蘭
- 亞太地區
- 中國
- 印度
- 日本
- 澳洲
- 韓國
- 拉丁美洲
- 巴西
- 墨西哥
- 阿根廷
- 中東和非洲
- 沙烏地阿拉伯
- 南非
- 阿拉伯聯合大公國
第9章:公司簡介
- Beckman Coulter
- Bio-Rad Laboratories
- Bio-Techne
- F. Hoffmann-La Roche
- Genomic health
- MDxHealth
- Merck KGaA
- Myriad Genetics
- OPKO Health
- QIAGEN
- Siemens
- Thermo Fisher Scientific
- Veracyte
The Global Prostate Cancer Biomarkers Market was valued at USD 4.7 billion in 2024 and is estimated to grow at a CAGR of 13.5% to reach USD 16.5 billion by 2034. The market growth is propelled by the rising number of prostate cancer cases, particularly in aging male populations, and the evolution of advanced diagnostics. Growing awareness of early screening, along with a rise in government-sponsored healthcare programs, is fueling adoption across primary care settings. The integration of next-generation diagnostics, such as artificial intelligence-driven assessments, liquid biopsies, and genomic analysis platforms, is enhancing early-stage detection while supporting personalized therapeutic decisions. Emerging economies are also increasing investments in health infrastructure, which is broadening access to precision-based testing methods. These biomarker-driven strategies are reshaping cancer care by offering better accuracy, reduced invasiveness, and faster diagnostic workflows.

Major industry contributors such as OPKO Health, Beckman Coulter, Bio-Rad Laboratories, and Bio-Techne are at the forefront of driving innovation in the prostate cancer biomarkers market. These companies are not only enhancing the accuracy and speed of detection through cutting-edge diagnostic platforms but also shaping the future of personalized cancer care. By developing non-invasive, highly sensitive biomarker assays and integrating them with AI-based diagnostic systems, they enable earlier identification of disease, better monitoring of progression, and more effective treatment planning.
| Market Scope | |
|---|---|
| Start Year | 2024 |
| Forecast Year | 2025-2034 |
| Start Value | $4.7 Billion |
| Forecast Value | $16.5 Billion |
| CAGR | 13.5% |
The protein biomarkers segment generated USD 2.4 billion in 2024 and is forecasted to reach USD 8.2 billion by 2034, growing at a CAGR of 13.1%. Their dominance is anchored in widespread clinical acceptance and their routine use for disease detection and progression tracking. Biomarkers in this category, which are easily measured through standard blood assays, remain the first-line diagnostic tools due to their proven reliability, cost-effectiveness, and strong correlation with prostate cancer risk. Ongoing research continues to refine these biomarker technologies, allowing them to reduce unnecessary invasive diagnostics while maintaining accuracy. New biomarker-based scoring tools are also improving risk stratification and patient selection for further testing, optimizing treatment pathways.
In terms of application, the screening and early detection segment held the highest share of the market in 2024, accounting for 40.2%. This is attributed to the growing emphasis on early diagnosis in both public health systems and individual clinical practices. Screening platforms are now designed for high-volume workflows and utilize blood or urine-based biomarker assays to deliver fast, reliable results with minimal processing. These solutions are highly suitable for routine health checks and are expanding rapidly in outpatient environments where fast turnaround and ease of use are critical. The convenience and accessibility of non-invasive tests are making them integral to prostate cancer prevention strategies globally.
North America Prostate Cancer Biomarkers Market held 42.5% share in 2024. The region benefits from advanced medical infrastructure, high rates of disease incidence, and accelerated pathways for regulatory approvals. The US and Canada continue to lead in adopting cutting-edge diagnostic platforms, driven by strong healthcare expenditure and rising public awareness campaigns. Continuous investment in research, a well-developed diagnostics ecosystem, and the presence of major market participants further support innovation in this region. This leadership position is expected to persist as demand grows for reliable, fast, and minimally invasive cancer diagnostics.
Companies such as Myriad Genetics, QIAGEN, Siemens, Veracyte, Genomic Health, F. Hoffmann-La Roche, MDxHealth, Thermo Fisher Scientific, and Merck KGaA are among the key players reshaping the competitive landscape. These include expanding their test portfolios through R&D investments and strengthening partnerships with oncology labs, academic institutions, and hospitals. Firms are leveraging advanced genomic and proteomic technologies to improve the sensitivity and specificity of their biomarker platforms. They're also entering strategic collaborations for global distribution, scaling up manufacturing, and securing regulatory approvals to speed up time-to-market. Many companies are focusing on companion diagnostics development, which ties directly into precision medicine, enhancing treatment alignment and patient outcomes.
Table of Contents
Chapter 1 Methodology and Scope
- 1.1 Market scope and definition
- 1.2 Research design
- 1.2.1 Research approach
- 1.2.2 Data collection methods
- 1.3 Data mining sources
- 1.3.1 Global
- 1.3.2 Regional/Country
- 1.4 Base estimates and calculations
- 1.4.1 Base year calculation
- 1.4.2 Key trends for market estimation
- 1.5 Primary research and validation
- 1.5.1 Primary sources
- 1.6 Forecast model
- 1.7 Research assumptions and limitations
Chapter 2 Executive Summary
- 2.1 Industry 3600 synopsis
- 2.2 Key market trends
- 2.2.1 Regional trends
- 2.2.2 Type trends
- 2.2.3 Application trends
- 2.2.4 End use trends
- 2.3 CXO perspectives: Strategic imperatives
- 2.3.1 Key decision points for industry executives
- 2.3.2 Critical success factors for market players
- 2.4 Future outlook and strategic recommendations
Chapter 3 Industry Insights
- 3.1 Industry ecosystem analysis
- 3.2 Industry impact forces
- 3.2.1 Growth drivers
- 3.2.1.1 Rising incidence of prostate cancer
- 3.2.1.2 Advancements in biomarker-based diagnostic technologies
- 3.2.1.3 Increased R and D investments from the public and private sectors
- 3.2.1.4 Growing adoption of personalized medicine through biomarker testing
- 3.2.2 Industry pitfalls and challenges
- 3.2.2.1 High cost of advanced diagnostic tests
- 3.2.2.2 Limited access and awareness in rural and underdeveloped regions
- 3.2.3 Market opportunities
- 3.2.3.1 Rising investments in non-invasive biomarker platforms
- 3.2.3.2 Increasing use of combination biomarker panels for early diagnosis
- 3.2.1 Growth drivers
- 3.3 Growth potential analysis
- 3.4 Regulatory landscape
- 3.5 Technological advancements
- 3.5.1 Current technological trends
- 3.5.2 Emerging technologies
- 3.6 Future market trends
- 3.7 Gap analysis
- 3.8 Porter's analysis
- 3.9 PESTEL analysis
Chapter 4 Competitive Landscape, 2024
- 4.1 Introduction
- 4.2 Company market share analysis
- 4.3 Company matrix analysis
- 4.4 Competitive analysis of major market players
- 4.5 Competitive positioning matrix
- 4.6 Key developments
- 4.6.1 Mergers and acquisitions
- 4.6.2 Partnerships and collaborations
- 4.6.3 New product launches
- 4.6.4 Expansion plans
Chapter 5 Market Estimates and Forecast, By Type, 2021 - 2034 ($ Mn)
- 5.1 Key trends
- 5.2 Genetic Biomarkers
- 5.3 Protein Biomarkers
- 5.4 Cell-based Biomarkers
- 5.5 Metabolomic Biomarkers
Chapter 6 Market Estimates and Forecast, By Application, 2021 - 2034 ($ Mn)
- 6.1 Key trends
- 6.2 Screening and early detection
- 6.3 Diagnosis and risk stratification
- 6.4 Prognosis and treatment monitoring
- 6.5 Companion diagnostics
Chapter 7 Market Estimates and Forecast, By End Use, 2021 - 2034 ($ Mn)
- 7.1 Key trends
- 7.2 Hospitals
- 7.3 Diagnostic laboratories
- 7.4 Cancer research institutes
- 7.5 Biopharmaceutical companies
Chapter 8 Market Estimates and Forecast, By Region, 2021 - 2034 ($ Mn)
- 8.1 Key trends
- 8.2 North America
- 8.2.1 U.S.
- 8.2.2 Canada
- 8.3 Europe
- 8.3.1 Germany
- 8.3.2 UK
- 8.3.3 France
- 8.3.4 Spain
- 8.3.5 Italy
- 8.3.6 Netherlands
- 8.4 Asia Pacific
- 8.4.1 China
- 8.4.2 India
- 8.4.3 Japan
- 8.4.4 Australia
- 8.4.5 South Korea
- 8.5 Latin America
- 8.5.1 Brazil
- 8.5.2 Mexico
- 8.5.3 Argentina
- 8.6 Middle East and Africa
- 8.6.1 Saudi Arabia
- 8.6.2 South Africa
- 8.6.3 UAE
Chapter 9 Company Profiles
- 9.1 Beckman Coulter
- 9.2 Bio-Rad Laboratories
- 9.3 Bio-Techne
- 9.4 F. Hoffmann-La Roche
- 9.5 Genomic health
- 9.6 MDxHealth
- 9.7 Merck KGaA
- 9.8 Myriad Genetics
- 9.9 OPKO Health
- 9.10 QIAGEN
- 9.11 Siemens
- 9.12 Thermo Fisher Scientific
- 9.13 Veracyte




